G Surgiwear Dental Drape Package (Sterile Dental Procedure Drapes)
Pack Sizes Available
Product Description
Technical Specifications
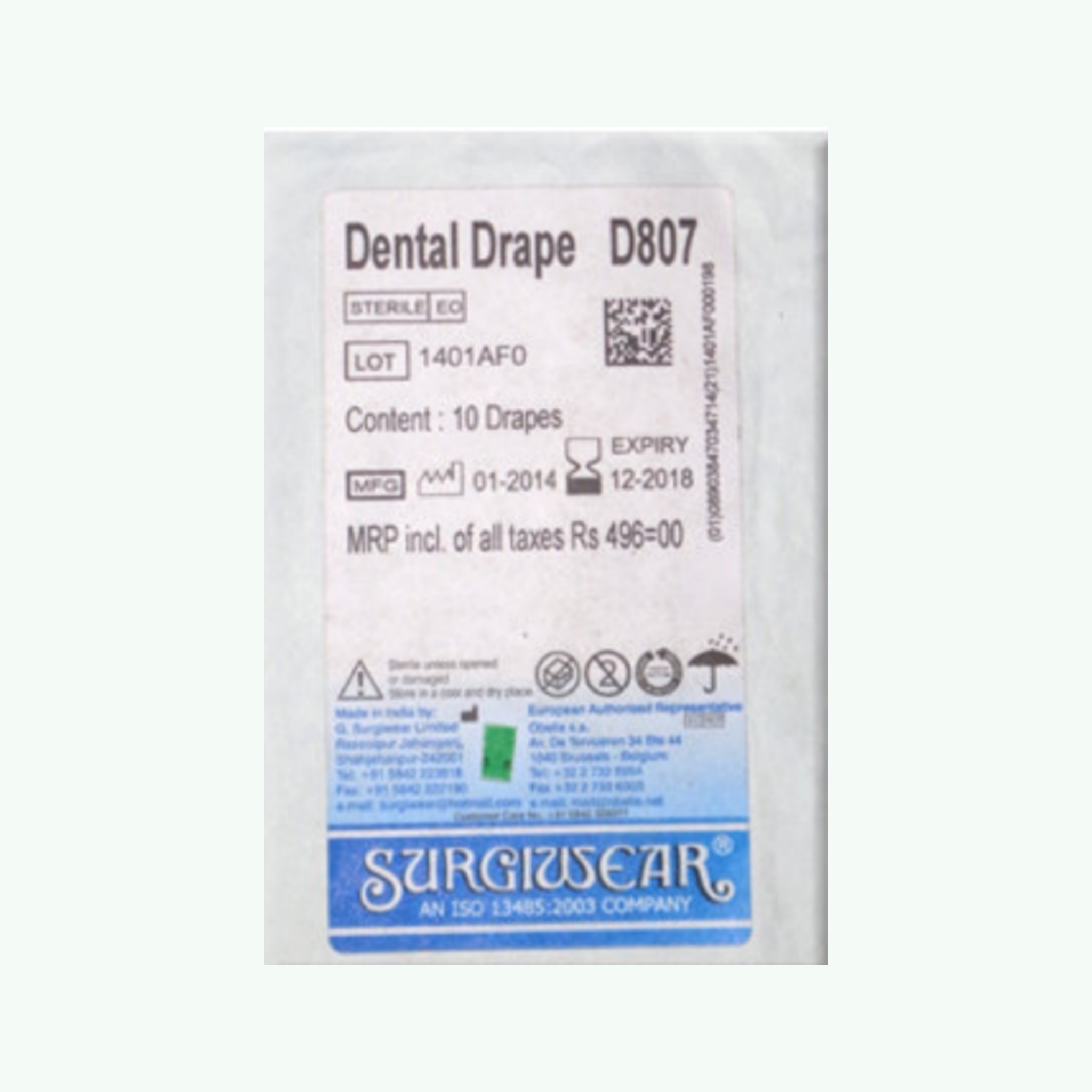
- Material: SMS (Spunbond-Meltblown-Spunbond) non-woven fabric — tri-layer fluid-resistant construction
- Sterility: Individually sterile wrapped — single-use disposable
- Pack Formats: 10 drapes per pack; 50 drapes per bulk institutional pack
- Configurations Available: Bibs with neck adhesive strip; procedure drapes; full-body barrier options (confirm at order)
- Primary Applications: Patient clothing protection during dental procedures; chairside barrier; aerosol splatter protection
- Manufacturer: G Surgiwear, India
Frequently asked questions
A dental bib (patient napkin) is typically a three-layer tissue-backed paper bib with a plastic backing, designed for routine chairside use to protect the patient's clothing from minor splatter and debris during routine dental procedures. A dental procedure drape is a larger format, typically made from non-woven fabric with better fluid resistance, designed to provide more comprehensive patient coverage and a better fluid barrier during more aerosol-generating procedures like ultrasonic scaling, air polishing, surgical extraction, or implant placement. The drape also provides more coverage area and may be used as part of a sterile field setup.
SMS stands for Spunbond-Meltblown-Spunbond — a three-layer non-woven fabric construction. The outer two spunbond layers provide strength, tear resistance, and a soft surface texture for patient comfort. The central meltblown layer consists of ultra-fine fibres that create a dense filtration matrix with high fluid barrier and particle retention properties. This tri-layer construction makes SMS fabric significantly more fluid-resistant than a single-layer spunbond non-woven, which is important for dental procedures involving water, blood, and aerosols generated by handpiece and ultrasonic instruments.
Dental procedure drapes protect the patient's clothing and exposed skin from direct aerosol splatter from handpieces and scalers. They do not protect the patient from inhalation of airborne aerosol — that requires respiratory protection such as masks and high-volume evacuation at the source. The patient-covering function of the drape is primarily for clothing protection and to provide a barrier against instrument or material drops landing on the patient's body. In a full infection control protocol, the dental drape is one component alongside high-volume evacuation, rubber dam isolation, and appropriate PPE for the operator.
Standard dental procedure drapes (such as the G Surgiwear dental drape) provide a clean barrier but should not be used as the primary sterile field in surgical procedures unless they are specifically classified and packaged as sterile surgical drapes meeting the relevant surgical textile standards. For oral surgery, implant placement, and periodontal surgery procedures requiring a sterile surgical field, purpose-made sterile surgical draping systems designed and validated for surgical use are the appropriate choice. Confirm the classification and sterility assurance level of any drape product before using it in a surgical application.
Dental procedure drapes contaminated with patient body fluids (blood, saliva) during procedures are classified as clinical waste — a regulated waste category in most jurisdictions. They should be disposed of in yellow clinical waste bags or containers designated for infectious/clinical waste, segregated from general office waste. The specific disposal classification and method should follow local health authority regulations for clinical waste management in dental settings. Uncontaminated drapes can be disposed of as general waste in some jurisdictions, but clinical assessment of each drape's contamination status should govern the disposal decision.
Related Products
Explore more high-quality items from our Hospital & Surgical Supplies range.

Air Bed FDA Certified (Alternating Pressure Bubble Mattress)
Nobody thinks about a mattress until the mattress becomes a problem. For most patients it never does. But for a person who is bedridden — whether from a stroke, a spinal injury, post-surgical recovery, or the long, slow immobility of advanced age — the mattress becomes everything. It is the surface against which the body presses, constantly, without relief. And without intervention, that constant pressure cuts off circulation to skin and underlying tissue, and a pressure ulcer develops. Quietly, painfully, and entirely preventably. The anti-decubitus alternating pressure bubble mattress — FDA and CE certified, medical-grade in construction — exists precisely to interrupt that cycle. The surface is made up of 130 individual air bubbles arranged in alternating groups. The electric pump inflates and deflates these groups on a timed cycle, typically every 6 to 10 minutes. As one set of bubbles inflates, the adjacent set deflates. The pressure points on the patient's body shift constantly and rhythmically, never allowing prolonged compression on any one area of skin. Blood flow continues. Tissue survives. Bedsores are prevented. The pump itself is built for continuous 24-hour operation. Noise levels sit below 35 dB on most variants — quiet enough for overnight use without disturbing the patient's sleep. The variable pressure dial on the pump allows caregivers to adjust firmness based on the patient's body weight, which directly affects how well the alternating cycle works. The mattress material is medical-grade PVC — waterproof, antibacterial, fire-resistant, and rated for patients up to 130 kg. The design fits standard hospital beds and home cots with adjustable side hangers. For healthcare equipment importers, medical retail chains, and hospital procurement managers — this is a product with strong, consistent demand. Ageing populations across the Middle East, Southeast Asia, and Africa are driving significant growth in home healthcare equipment. Every nursing home, every rehabilitation unit, every ICU in a country with a growing elderly population needs these mattresses. The procurement cycle is not one-off; facilities replace and expand their mattress stock regularly. Sara Wellness supplies medical air beds and home healthcare equipment to wholesale buyers across multiple international markets. We are not a retail channel. We work with importers and distributors who are building product lines for hospital group supply or retail healthcare chains. Our 15 years of export experience means we understand how medical equipment import documentation, certification requirements, and voltage specifications vary from market to market. We'll discuss all of it before the order is placed, not after. MOQs are negotiable. Bulk carton pricing improves considerably when orders combine air beds with related home healthcare equipment SKUs. If you are sourcing for the home healthcare or hospital supply market, this is a product worth adding to the conversation.

Bellcross N95 Particulate Filter Respirator and Surgical Mask
The N95 mask became a household conversation topic during a global pandemic. Before that, it was simply the thing the ICU nurse reached for before entering an isolation room, the thing the construction worker wore cutting drywall, the thing the tuberculosis ward always had in stock. The Bellcross N95 Particulate Filter Respirator is a product that sits at the intersection of both worlds — healthcare-grade protection from a manufacturer with four decades of disposable medical product manufacturing experience in India. N95 means exactly what it says. Ninety-five percent filtration efficiency for non-oil-based particulate matter when properly fitted to the face. This is the threshold established by NIOSH (National Institute for Occupational Safety and Health) — the US regulatory authority for respiratory protection in occupational settings. An N95 respirator does not rely on loose fit or general aerosol reduction. It relies on a tight facial seal that forces all inhaled air through the filter medium, and the filter medium that captures at least 95% of particles 0.3 microns and larger (the most penetrating particle size for fibrous filter materials). The Bellcross version serves dual purpose as both a particulate respirator and a surgical mask — providing the fluid resistance layer that distinguishes a healthcare surgical N95 from a standard industrial N95. The fluid-resistant outer layer adds protection against splash and spatter of blood and body fluids, which the NIOSH-only industrial N95 standard does not address. For healthcare settings, this is the variant that makes clinical sense — particularly in operating theatres, procedure rooms, endoscopy units, and any clinical area where aerosol-generating procedures are performed. Bellcross Industries, established in 1984 in Mumbai, is one of India's largest manufacturers and exporters of disposable medical products. The range covers surgical drapes, OT gowns, HIV safety kits, ECG electrodes, and the full spectrum of single-use hospital consumables. The N95 respirator is manufactured to the same quality standards that have made Bellcross products trusted across export markets for over four decades. For PPE distributors and hospital supply importers sourcing respiratory protection from India — Bellcross N95 respirators offer authentic Indian manufacturing credentials, not re-labelled imports. Sara Wellness exports Bellcross disposable medical products to international wholesale buyers. Flexible MOQs, direct one-on-one dealings, and fifteen years of export experience behind every shipment.

Bellcross Disposable HIV Safety Kit (Complete PPE Set — Infection Control)
The HIV safety kit was born from a straightforward clinical recognition: the risk of occupational blood-borne pathogen exposure is not random, and it is not limited to surgeons. It is nurses doing IV insertions. It is phlebotomists drawing blood on patients whose infection status is not yet confirmed. It is emergency department staff dealing with a trauma patient before serology results are back. The kit puts the complete barrier protection ensemble together in one pre-assembled, pre-sterilised pack that can be grabbed and donned in under two minutes. Bellcross Disposable HIV Safety Kits contain everything a healthcare worker needs to create a complete bodily fluid barrier before attending to a patient where blood-borne pathogen exposure risk exists. The gown is full length, full sleeves, with elastic cuffs and a PVC apron interlining that makes it fluid-impermeable rather than merely fluid-repellent. The goggles cover the periocular area — because blood splash to the conjunctiva is a documented HIV transmission route. The shoe covers close off the lower extremity pathway. The gloves close off the hand pathway. The biohazard waste bag is included because the kit itself, when removed, is clinical waste. Bellcross manufactures both sterile (ETO sterilised with indicator) and non-sterile kit variants. The sterile format is appropriate for procedural settings where the kit contents may enter or work adjacent to a sterile field. The non-sterile format is the more common clinical procurement choice for general barrier precaution use. Kits can be customised to buyer specification — the contents list above represents the standard Bellcross configuration, but institutional buyers who have different PPE standards, local regulatory requirements, or specific gown specifications can request modifications at order. Bellcross Industries Pvt. Ltd., established in 1984, manufactures every component of the HIV safety kit in-house from its Mumbai facility. This is not a kit assembled from third-party components — the gown, mask, cap, goggles, shoe covers, and bag are all Bellcross manufacture. The supply chain consistency this provides matters for institutional buyers who need to know what is inside every sealed pack. Sara Wellness exports Bellcross HIV safety kits to international wholesale and institutional buyers. Carton quantities, custom configurations, and fifteen years of export coordination on every order.