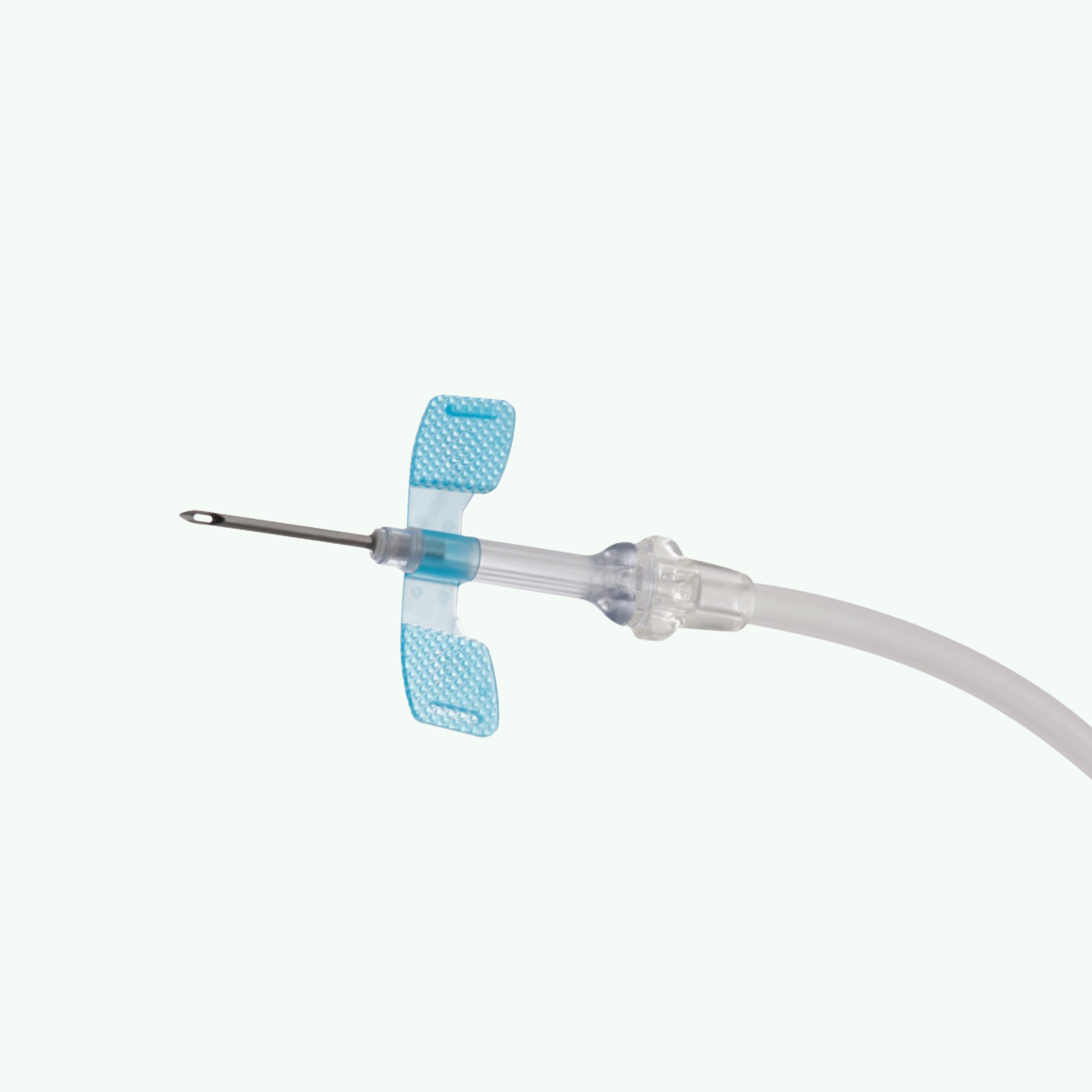
AVF Needles 16G 50's Pack (Nipro)
Three times a week. That's the rhythm of a hemodialysis patient's life. Three times a week, a needle goes into their fistula, blood is drawn out, cleaned by a machine, and returned. It's a procedure measured in hours and repeated hundreds of times over the course of a patient's treatment. When something is done that often, the margin for error is zero, and the tolerance for poor equipment is even lower. The Nipro Standard AVF (Arteriovenous Fistula) Needle, 16G, is the needle that dialysis centres across the world choose for patients whose blood flow rate runs between 300 and 350 ml/min. That gauge-to-flow-rate relationship is not arbitrary. Nipro's own clinical guidance and the broader nephrology literature both point to matching needle gauge to patient flow to protect fistula integrity over years of repeated access — and the 16G sits in a range where the balance between access size and vessel preservation is optimal for a large proportion of the dialysis population. The needle itself is built around three engineering decisions that nephrologists and dialysis nurses notice immediately. First, the ultra-sharp siliconized 3-bevel tip punctures cleanly with minimal vessel trauma. Second, the ultra-thin cannula wall means the outer gauge is as small as possible for a given inner diameter, again protecting the vessel. Third, the oval-shaped back-eye on the arterial needle optimises blood flow while minimising the risk of vessel wall aspiration — a complication that causes localised trauma at every session if present and absent when the back-eye geometry is correct. The turnable wings give the cannulating nurse the ability to adjust needle positioning after insertion, which matters in patients with awkward fistula anatomy. The 300mm tubing provides comfortable connection to the blood circuit without tension on the cannulation site. Sterilization is dual-method: ETO and gamma, confirming compliance with global hospital sterility standards. For dialysis supply importers, wholesale distributors, and hospital procurement teams across Africa, the Middle East, Southeast Asia, and South Asia — Nipro is a name that dialysis unit managers already trust. You're not selling an unknown brand. You're restocking a product that the clinical team has already formed an opinion about, which is the easiest sell in any B2B supply chain. Sara Wellness supplies Nipro dialysis consumables in pack-of-50 retail units and 500-unit wholesale cartons. Both formats move well across the markets we serve. Our 15 years of dialysis supply export experience means we know which documentation dialysis-specific procurement requires, what shelf life expectations look like per market, and how to structure a combined order across multiple gauge sizes to maximise your order value. Reach out with your gauge mix and volume requirement, and we'll respond with a real quote from a real person.
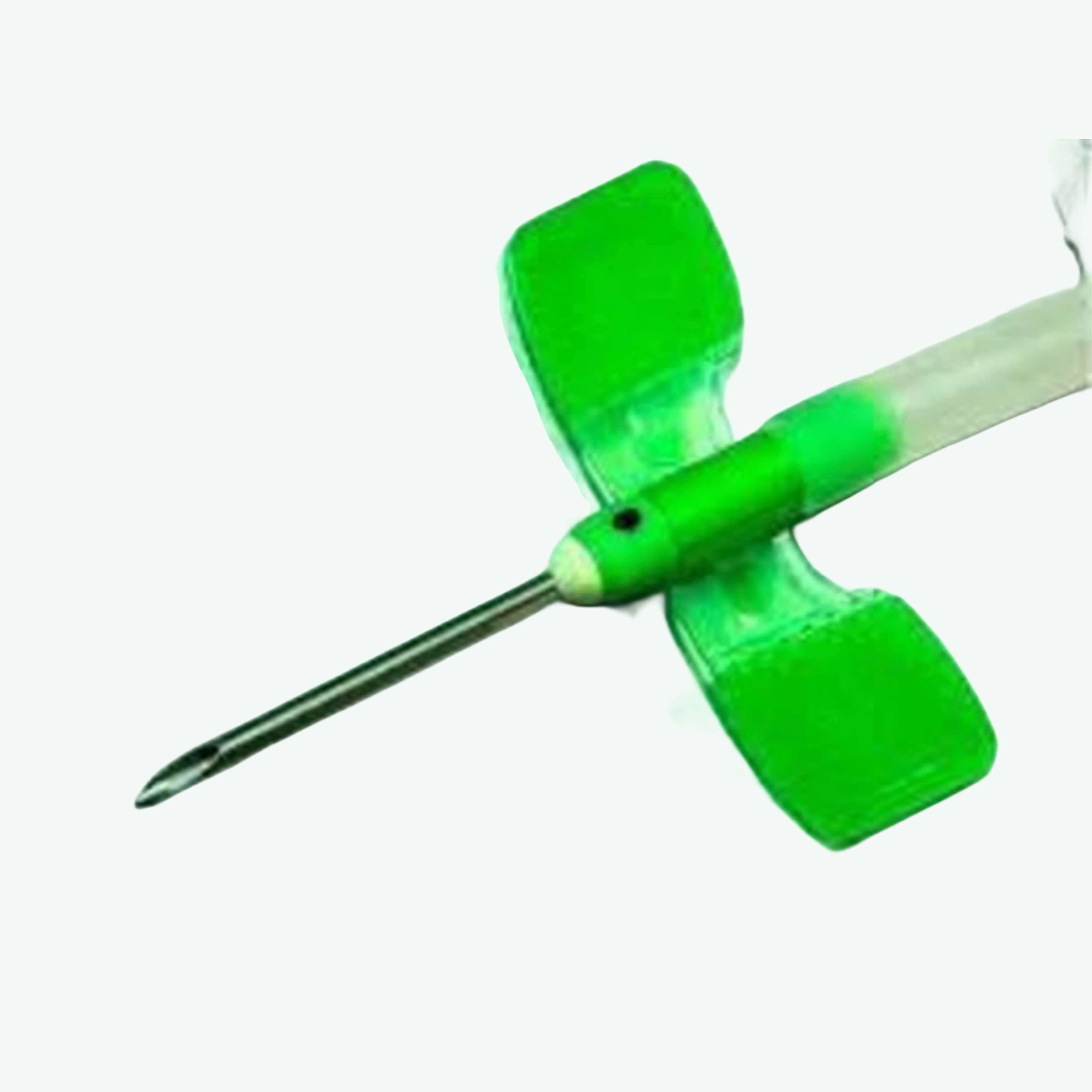
AVF Needles 17G 50's Pack (Nipro)
In nephrology, the 17G AVF needle has earned itself a very specific place in the clinical hierarchy. It's the needle that earns a fistula's trust. When a new arteriovenous fistula is formed — surgically constructed, carefully matured over weeks, finally declared ready for use — the first few cannulations are the most consequential. Too large a needle on a young fistula and you risk trauma that can compromise access that a patient may depend on for years or decades. Nipro's own clinical guidance, consistent with published nephrology literature, explicitly recommends starting with a 17G for the first sessions. That's not a small clinical detail. For dialysis unit managers, procurement officers, and clinical leads choosing which needles to stock, knowing that the 17G serves a specific and irreplaceable role in fistula maturation protocols actually drives purchasing decisions in a way that pure price comparison never does. Nipro's Standard AVF Needle in 17G is colour-coded yellow across all markets — a universal convention that allows dialysis nursing staff to identify gauge at a glance without reading packaging during a busy session. The textured wings are colour-matched and provide a secure, grippy surface for taping after placement. Turnable wings give the cannulating nurse the flexibility to position the wing tabs flat against the skin in the most anatomically comfortable orientation after insertion, which matters during long, four-hour sessions. The 3-bevel siliconized tip enters cleanly and with minimal resistance. The oval back-eye on the arterial needle is designed to prevent vessel wall aspiration at the blood flow rates typical for this gauge. Tube length runs to 300mm standard, with the Luer-lock connector and clamp included per unit. For importers and wholesale distributors building out dialysis consumable lines — the 17G is non-negotiable to stock alongside the 15G and 16G. A dialysis centre that orders one gauge from you will need the others. Gauge-complete ordering is how most dialysis unit procurement works, and Sara Wellness can supply across the full Nipro fistula needle range in a single order. Fifteen years of dialysis supply exports have shown us that this category rewards reliability above all else. Dialysis is not an elective service. Centres cannot run out of fistula needles. That reality shapes how we approach every order — with proper lead time conversations, realistic stock commitments, and documentation that clears customs the first time. If you're sourcing Nipro AVF needles for distribution across Africa, Southeast Asia, or the Middle East, talk to us before you talk to anyone else. The conversation is free and the quote will be honest.

Adfusion Hemodialysis Blood Tubing Set
Hemodialysis is a procedure with a very low tolerance for equipment failure. Blood leaves the body, travels through a machine, and returns — cleaner, corrected, rebalanced. That extracorporeal circuit is kept alive by tubes, clamps, chambers, and connectors that need to perform predictably for three to five hours, three times a week, for years. One weak joint, one collapsed drip chamber, one clamp that doesn't hold — and the session is compromised before the first hour is done. The Adfusion Hemodialysis Blood Tubing Set is a complete bloodline set — arterial and venous lines — designed for exactly the kind of session-after-session reliability that dialysis units expect and depend on. The arterial line draws blood from the patient's vascular access point to the dialyzer inlet. The venous line returns cleaned blood from the dialyzer outlet back to the patient. The drip chambers along both lines are engineered to reduce foaming and improve air elimination during priming — two of the most common setup complications in busy, high-throughput dialysis centres. Colour-coded ergonomic clamps identify arterial (red) and venous (blue) lines at a glance, reducing the risk of line mix-up during preparation and reconnection. Kink-resistant tubing maintains flow integrity even when lines are repositioned or stretched across a patient's arm during the session. The tubing itself is medical-grade PVC, non-toxic, pyrogen-free, and EO sterilized to the standards expected in clinical dialysis environments. Compatibility is universal. The Adfusion set is designed and confirmed for use with Fresenius, B. Braun, Campbell, Baxter, and Japan-brand hemodialysis machines, covering the machine mix found in most dialysis centres across the Middle East, Africa, and Southeast Asia. Dialysis units running mixed machine fleets — which is more common than procurement managers usually advertise — can stock a single tubing set SKU rather than managing multiple SKU lines per machine brand. For importers and wholesale distributors, blood tubing sets are the highest-volume consumable in any dialysis supply chain. Every patient requires a new set for every session. There is no sharing, no reuse, no stretching a box across more sessions than it contains. This makes tubing sets the SKU that dialysis distributors reorder most frequently and plan most carefully. Getting your supplier right for this product matters more than it does for almost anything else in the dialysis category. Sara Wellness has been exporting dialysis consumables to buyers across Africa, the Middle East, and Southeast Asia for 15 years. We understand high-frequency ordering, we understand the documentation dialysis-specific imports require, and we handle every account with the kind of direct communication that makes reordering straightforward. MOQs are negotiable. Volume pricing improves meaningfully when you combine blood tubing sets with AVF needles and transducer protectors in a single order.

Arrow Acute Hemodialysis Catheter Kit (Teleflex — Double Lumen)
There is a particular sequence of decisions that happens in a dialysis unit when a patient's existing vascular access fails. The fistula is not maturing. The graft is thrombosed. The tunnelled catheter has an exit site infection. The patient has a scheduled dialysis session in six hours. What happens next — who places what, how quickly, and with what device — determines whether that session happens on time. The Arrow Acute Hemodialysis Catheter Kit is the device that provides the temporary vascular access that makes dialysis possible in exactly those situations. It is a large-bore, double-lumen, polyurethane catheter designed for percutaneous insertion into the internal jugular, subclavian, or femoral vein using the Seldinger technique — the guidewire-facilitated approach that has been the standard for central venous catheter placement for six decades. The two lumens — arterial and venous, colour-coded red and blue — create the separate inflow and outflow circuit that the hemodialysis machine requires to complete the extracorporeal blood circuit. The Arrow brand is manufactured by Teleflex — one of the most recognised names in vascular access globally. The catheter is available with ARROWg+ard antimicrobial technology, which incorporates chlorhexidine and silver sulfadiazine coating to reduce the risk of catheter-related bloodstream infection (CRBSI) — a clinically significant complication in dialysis patients who are already immunocompromised and require repeated and prolonged catheter access. CRBSI prevention is a major procurement consideration for hospital infection control committees across every market. Sizes range from 12Fr (for patients with smaller veins or when a smaller profile is clinically preferred) through 13Fr and 14Fr in lengths of 15cm, 20cm, and 24cm, corresponding to the appropriate length for right internal jugular, left internal jugular, and femoral insertion sites respectively. The soft catheter tip balances insertion capability with indwelling performance — firm enough to track over the guidewire without buckling, soft enough to reduce vessel wall trauma during dwelling. For dialysis supply distributors and hospital procurement managers in nephrology and critical care supply chains — the Arrow acute hemodialysis catheter is a device where brand recognition, clinical familiarity, and FDA/CE regulatory clearance combine to make procurement straightforward. Nephrologists and critical care physicians have brand preferences in vascular access, and Arrow is one of the top two or three global references. Sara Wellness exports Teleflex Arrow dialysis catheters and related vascular access products to wholesale buyers internationally. Pack of 1 per sterile kit. Contact us with size specification and volume requirement.

Dialysis Acid Concentrate (Hemodialysis Concentrate Part A — Liquid)
Dialysis is not a treatment that happens in the machine. It happens in the dialysate — the precisely formulated electrolyte solution that flows counter-current to the patient's blood across a semipermeable membrane, drawing out waste products and excess fluid while maintaining the patient's electrolyte and acid-base balance. Get the dialysate composition right, and the patient comes off the machine with stable potassium, corrected bicarbonate, and appropriate fluid balance. Get it wrong, and the consequences are immediate and serious. Dialysis Acid Concentrate — Part A — is one of the two components that the hemodialysis machine mixes with purified water to produce dialysate. The acid concentrate (liquid) contains the essential electrolytes: sodium chloride, calcium chloride, potassium chloride, magnesium chloride, dextrose, and acetic acid or citric acid, formulated in precise concentrations. The acetic acid component serves as the acidifying agent that prevents calcium carbonate precipitation when the concentrate is mixed with bicarbonate, and contributes to the acetate-buffered portion of the dialysate composition. Part B (sodium bicarbonate) provides the primary buffer system. The dialysis machine proportions both concentrates with purified water to produce dialysate at a fixed ratio — typically 1:34 or 1:44 depending on the machine and concentrate specification. The formulation follows European, British, and United States Pharmacopoeia standards for hemodialysis concentrates. Electrolyte concentrations in the ready-to-use dialysate are calibrated to physiological targets: sodium 136-140 mmol/L, potassium 2.0-2.5 mmol/L, calcium 1.25-1.75 mmol/L, magnesium 0.5 mmol/L, chloride approximately 103 mmol/L, acetate 4-4.5 mmol/L, and bicarbonate approximately 35 mmol/L. The concentrate is supplied in sealed 5-litre and 10-litre jerry cans — the standard supply format for dialysis centres across South Asia, Africa, and the Middle East. Labels are marked red as per international convention for acid concentrate (Part A) to prevent confusion with Part B bicarbonate (typically labelled blue or white). For dialysis supply distributors and pharmaceutical export buyers — hemodialysis acid concentrate is a foundational consumable. Every hemodialysis session at every machine consumes one proportioned volume of acid concentrate. The consumption rate across a dialysis centre with multiple machines running multiple sessions per day is substantial and entirely predictable. Sara Wellness exports hemodialysis dialysis concentrates and related consumables with proper cold-chain and hazardous goods documentation where applicable.

Fresenius Blood Tubing Set (Hemodialysis — Arterial and Venous Bloodlines)
Three times a week, for four hours at a time. That is the schedule that defines the life of an end-stage renal disease patient on haemodialysis. Every session requires a new blood tubing set — arterial and venous bloodlines that connect the patient's vascular access to the dialysis machine and back, forming the extracorporeal circuit through which approximately 300ml of blood at any moment circulates outside the body. This is not a product category with room for unreliability. Fresenius Blood Tubing Sets are manufactured by Fresenius Medical Care, the world's largest integrated provider of dialysis products and services. The CombiSet True Flow series — Fresenius's primary bloodline range — is designed with a specific engineering focus: accurate and consistent blood flow rate from the first minute of treatment to the last. The polymer blend and extrusion technology used in CombiSet bloodlines is formulated to maintain pump segment compliance consistently, so the blood flow rate displayed on the machine accurately reflects actual flow rather than drifting as the tubing fatigues during a four-hour session. The Twister reverse flow device (included in selected CombiSet configurations) addresses one of the most maintenance-intensive dialysis machine monitoring requirements: monthly blood flow measurement verification. Conventionally, this requires manually disconnecting the bloodlines — a blood exposure risk. The Twister's closed-system design allows flow reversal and measurement without disconnection, eliminating blood contact risk for the dialysis technician performing the check. Key design features across the Fresenius bloodline range include: colour-coded clamps for arterial vs. venous identification (a patient safety feature preventing line transposition); anti-foaming drip chambers; clear transducer protectors for monitoring while meeting CMS guidelines; and multiple venous medication ports for flexible drug delivery during dialysis. All components that contact blood are medical-grade PVC formulated to minimise risk of haemolysis and complement the machine's pressure monitoring capabilities. For dialysis consumable distributors and nephrology department procurement managers — Fresenius bloodlines are the native consumable for Fresenius 2008 series machines, which represent a significant proportion of the global haemodialysis machine installed base. Sara Wellness exports Fresenius dialysis consumables to international wholesale buyers.

Fresenius 4008S Hemodialysis Machine
Dialysis centres around the world make purchasing decisions with an unusual level of scrutiny. The machine that sits in a treatment bay runs sessions three or more times a week, per patient, indefinitely. It collects data, manages alarms, handles ultrafiltration volumes, and keeps a patient alive during a session that takes four hours. A decision that goes wrong here costs more than money. The Fresenius 4008S is one of the most recognisable hemodialysis platforms in the world, and its reputation rests on the same foundation as most durable medical equipment: it has been around long enough for the market to have formed a very clear view of it. The 4008 product family has been in clinical use for over 25 years, deployed across dialysis centres in Europe, Asia, Africa, and the Americas. The 4008S specifically is the mid-range variant in that family, positioned to deliver high-quality standard hemodialysis without the premium pricing of the more advanced 5008 platform. The machine features a 10.4-inch TFT-LCD monitor displaying treatment parameters graphically, which reduces misreading of critical values during a session. The volumetrically controlled ultrafiltration system provides precise fluid removal management — the element of hemodialysis most directly connected to haemodynamic stability during the session. The integrated Blood Pressure Monitor simplifies vital signs monitoring without requiring a separate device at each chair. The Online Clearance Monitor (OCM) is the 4008S's most clinically significant optional feature. OCM measures dialysis adequacy continuously and in real time during the session, tracking effective urea clearance (K), accumulated Kt, delivered Kt/V dose, and plasma sodium concentration. This continuous monitoring replaces periodic manual checks and removes the lag in detecting inadequate treatment. The DIASAFEplus inline endotoxin filter enables the production of ultrapure dialysate fluid, meeting ISO 23500:2019 purity standards. Ultrapure dialysate is increasingly considered a baseline clinical standard in modern hemodialysis rather than an option. The 4008S dimensions are approximately 1370mm high x 500mm wide x 650mm deep with a weight of approximately 80-86kg. It runs on 230V, 50/60Hz. Sara Wellness works with healthcare facility procurement offices and dialysis equipment distributors internationally. We handle export logistics and documentation with 15 years of medical export experience behind us.
Fresenius F6 HPS Polysulfone Low-Flux Dialyzer
The F6 is the workhorse of the Fresenius F-series HPS range. If you look at the global distribution of Fresenius dialyzer deployments across dialysis centres, the F6 is consistently the highest-volume model — not because it is the most technically advanced, but because 1.3 m² of membrane with the performance parameters of Fresenius Polysulfone happens to hit the clinical sweet spot for the average adult dialysis patient in the majority of standard three-times-weekly regimens. It is the size that achieves adequate Kt/V in most patients without being unnecessarily large, and its priming volume of 82 mL is manageable for most patients without causing haemodynamic instability at session start. The F6 HPS dialyzer uses the same Fresenius Polysulfone membrane and INLINE steam sterilisation as the rest of the F-series HPS range. The ultrafiltration coefficient of the F6 HPS is approximately 13 mL/h x mmHg — slightly higher than the F5, reflecting the increased membrane surface. The urea clearance at a blood flow rate of 200 mL/min and dialysate flow of 500 mL/min is approximately 186 mL/min; at 300 mL/min blood flow it reaches approximately 222 mL/min, which is adequate for achieving Kt/V targets of 1.2-1.4 in most standard adult patients. The microundulated fibre structure — the signature design of Fresenius Polysulfone — maintains uniform dialysate flow distribution around each hollow fibre, preventing the localised dialysate stagnation zones that reduce effective membrane utilisation in dialyzers with straight, non-undulating fibres. This structural feature is credited with the consistently high in-vitro-to-in-vivo performance correlation that clinical teams report with the F-series. For dialysis supply procurement teams, the F6 HPS is the standard dialyzer specification for adult standard HD protocols. It is the model against which alternative dialyzer proposals are most commonly benchmarked. Sara Wellness exports Fresenius dialysis products internationally.

Fresenius Hemoflow F5 HPS Low-Flux Polysulfone Dialyzer
In a dialysis unit running twenty sessions a day, the dialyzer is the consumable that defines the clinical chemistry of each treatment. The membrane material, the pore architecture, the surface area, the ultrafiltration coefficient — all of these determine what molecular species are removed from the patient's blood and at what rate. Choosing the right dialyzer for each patient category is a clinical decision. Getting it reliably, consistently, and without supply chain disruption is a procurement decision. The Fresenius Hemoflow F5 HPS is a low-flux polysulfone dialyzer in the F-series HPS (High Performance Steam) range from Fresenius Medical Care — one of the world's most widely used dialyzer product lines. The F5 identifies a specific surface area configuration: 1.0 m² of Fresenius Polysulfone membrane, appropriate for smaller adult patients and patients with lower body surface area who require adequate dialysis dose (Kt/V) without overshooting with a larger-surface dialyzer. Fresenius Polysulfone is the proprietary membrane material that has been the benchmark for synthetic dialysis membranes since its introduction. Its high endotoxin retention capability is attributed to the chemical composition and the special undulating (microundulated) hollow fibre structure — the wavy fibre geometry prevents dialysate channelling around the fibre bundle, ensuring that dialysate contacts each hollow fibre uniformly and solute transport occurs at maximum efficiency across the full membrane surface. The HPS designation indicates INLINE Steam sterilisation — the F-series HPS dialyzers are sterilised by direct steam injection rather than by chemical agents (ethylene oxide) or gamma irradiation. This produces a dialyzer that is completely free of sterilisation residues, is pyrogen-free, and has biocompatibility characteristics that are not modified by the sterilisation process. The ultrafiltration coefficient of the F5 HPS is approximately 10 mL/h x mmHg. Sara Wellness exports Fresenius dialysis products to medical distributors internationally.

Fresenius Polysulfone F8 HPS Low-Flux Dialyzer
When a dialysis centre receives a large patient — a patient with a body surface area in the upper quartile, a patient on a high-clearance protocol, or a patient where the previous session's Kt/V calculation keeps coming out short despite optimised blood and dialysate flow rates — the clinical team reaches for the largest effective membrane area in their standard stock. The Fresenius F8 HPS is that dialyzer: 1.8 m² of Fresenius Polysulfone membrane, built on the same design principles as the rest of the F-series HPS but scaled to the highest surface area in the standard HPS configuration. At 1.8 m², the F8 delivers urea clearance of approximately 240 mL/min at blood flow rates of 400 mL/min — the high end of standard blood pump operation. The priming volume of approximately 113 mL is the largest in the standard HPS range, which means the extracorporeal circuit blood volume is highest with this dialyzer. For patients with cardiovascular compromise or poor haemodynamic stability, this should be taken into account in session management — particularly at session commencement when the priming volume of saline is replaced with blood. The F8 HPS shares the INLINE steam sterilisation and microundulated Fresenius Polysulfone membrane of the F5 and F6 models. The ultrafiltration coefficient is approximately 18 mL/h x mmHg — at the higher end for a low-flux dialyzer, which requires appropriate transmembrane pressure management to achieve the target ultrafiltration rate without inadvertent fluid removal overshoot. For dialysis supply procurement teams, the F8 is a standard line item alongside the F5 and F6, covering the high-body-mass or high-clearance-target patient population. Having the full F-series range available in the supply chain allows clinical teams to match dialyzer surface area to patient need without substituting different membrane technologies or sterilisation methods. Sara Wellness exports Fresenius dialysis products to distributors internationally.