
Microlisa HIV Ag & Ab 4th Generation ELISA Test Kit
Blood screening exists in a category of laboratory work where the margin for error is not just professionally unacceptable but medically catastrophic. A false negative in HIV screening does not just fail a test. It compromises patient safety, undermines transfusion protocols, and exposes healthcare systems to risks that nobody wants to calculate. Microlisa HIV Ag & Ab 4th Generation ELISA was engineered to close the detection window that makes early HIV infection so difficult to identify reliably. This is an in-vitro qualitative enzyme immunoassay designed for simultaneous detection of antibodies to HIV-1 (including Group O and subtype C prevalent in India), HIV-2, and HIV-1 p24 antigen in human serum or plasma. The test is intended for screening of blood donors, diagnostic testing of individuals at risk for HIV infection, and clinical evaluation of patients with AIDS-related symptoms. It represents the fourth generation of HIV ELISA technology, which detects both antibodies and antigens simultaneously rather than antibodies alone. The clinical advantage of 4th generation testing is the shortened window period. Traditional antibody-only tests miss early seroconversion cases where HIV-1 p24 antigen is present but antibodies have not yet developed to detectable levels. By detecting p24 antigen during the acute infection phase (typically 2 to 4 weeks post-exposure), this assay identifies infections approximately 1 to 2 weeks earlier than 3rd generation antibody-only tests. That earlier detection matters critically in blood donor screening and post-exposure monitoring. The assay is based on sandwich ELISA methodology. Microtiter wells are pre-coated with HIV envelope proteins (gp41, C-terminus of gp120 for HIV-1, and gp36 for HIV-2) and anti-p24 monoclonal antibodies. When specimens are added, any HIV antibodies or p24 antigen present bind to the coated antigens or antibodies. After washing, horseradish peroxidase (HRPO) conjugated antigens and anti-p24 antibodies are added, forming a sandwich complex. The colorimetric reaction develops proportionally to the amount of HIV antibodies or antigen present, read at 450nm absorbance. The kit uses color-coded reagents to monitor procedural steps, reducing protocol errors during multi-step workflows. Breakaway microwell strips allow testing flexibility from single specimens to full 96-well plate runs. Storage stability is maintained at 2-8°C with a shelf life of 24 months unopened. Total assay time including incubation steps is approximately 120 minutes. Sensitivity and specificity meet international standards for 4th generation HIV screening. Clinical evaluations demonstrate 100% sensitivity in detecting seroconversion panels and p24 antigen standards quantified down to 200 pg/ml. Specificity exceeds 99.5% when tested against large sample populations. The test detects all major HIV-1 subtypes including Group O and subtype C, which are epidemiologically significant in the Indian subcontinent. For distributors supplying blood banks, transfusion centers, diagnostic laboratories, and public health screening programs, Microlisa HIV Ag & Ab represents a clinically validated 4th generation screening platform with predictable reorder cycles. Sara Wellness has been exporting in-vitro diagnostic kits and laboratory reagents from India for 15 years.

Advantage PAN Malaria Card Rapid Diagnostic Test Kit
Malaria diagnosis in endemic regions operates under time pressure that microscopy cannot always accommodate. A patient presenting with fever in a rural health center at midnight does not have the luxury of waiting until morning for a trained microscopist to arrive, prepare slides, and spend twenty minutes examining blood films under oil immersion. That delay can mean the difference between timely artemisinin treatment and cerebral malaria developing overnight. Advantage PAN Malaria Card was designed to deliver species-level diagnosis in settings where microscopy is impractical or unavailable. This is a rapid visual immunoassay for qualitative detection of all four human Plasmodium species (P. falciparum, P. vivax, P. malariae, P. ovale) based on pan-specific plasmodium lactate dehydrogenase (pLDH) antigen in whole blood. The test provides results within 20 minutes using a simple fingerstick blood sample, no laboratory equipment required, making it ideal for point-of-care testing in primary health centers, rural clinics, field hospitals, and outbreak response settings. The assay is based on sandwich immunochromatography using monoclonal antibodies specific to pLDH, an enzyme produced by all Plasmodium species during their erythrocytic life cycle. When infected blood is added to the test device and assay buffer is applied, red blood cells lyse and pLDH antigen (if present) binds to gold-conjugated anti-pLDH antibodies. This complex migrates along the nitrocellulose membrane and is captured by immobilized anti-pLDH antibodies at the test line, producing a visible pink-purple band that confirms malaria infection. The see-through device design allows direct visualization of sample migration and result development, which helps identify invalid tests caused by insufficient sample volume or improper application. This transparency reduces the ambiguity that plagues some lateral flow devices where internal workings are hidden. Sensitivity and specificity have been validated through WHO malaria RDT evaluation programs using panels of wild and cultured parasites. The test detects parasitemia levels above 100 parasites per microliter of blood for both P. falciparum and P. vivax, which is clinically relevant for symptomatic infections requiring treatment. Specificity exceeds 99% when tested against cross-reactive conditions including dengue, leptospirosis, typhoid, and other febrile illnesses common in malaria-endemic areas. Shelf life is 24 to 30 months when stored at 4-30°C, which is critical for stockpiling in tropical climates where cold chain infrastructure is unreliable. The extended temperature stability means the test remains functional even when stored at ambient temperatures in resource-limited settings. Each kit contains individually sealed test devices, buffer vials, blood collection pipettes, and instructions for use. The test requires no special training beyond basic clinical skills and can be performed by nurses, paramedics, or trained community health workers. For distributors supplying national malaria control programs, public health departments, NGO field operations, and private diagnostic laboratories, Advantage PAN Malaria Card represents a WHO-evaluated rapid diagnostic platform with predictable consumption tied to malaria case loads. Sara Wellness has been exporting rapid diagnostic test kits and laboratory reagents from India for 15 years.

Human Serum Coombs Antisera (Antihuman Globulin Reagent)
Blood banking operates on a fundamental requirement that most people never think about until something goes wrong. Every unit of blood transfused must be confirmed compatible with the recipient's immune system. Every pregnant woman screened for antibodies that could harm her unborn child. Every suspected case of hemolytic anemia investigated for antibodies attacking the patient's own red blood cells. None of this happens without Coombs antisera making the invisible antibodies visible. Human Serum Coombs Antisera is the reagent that makes antiglobulin testing possible in blood banks and immunohematology laboratories worldwide. This is antihuman globulin (AHG) reagent used in both direct and indirect antiglobulin tests (Coombs tests) to detect antibodies and complement components bound to red blood cell surfaces or present free in serum. The reagent is produced by immunizing animals (typically rabbits) with human immunoglobulins, which induces production of polyclonal antibodies specific for human IgG antibodies and complement factor C3d. When added to washed red blood cells coated with IgG or complement, the antihuman antibodies bind to the human antibodies and form bridges between adjacent sensitized cells, causing visible agglutination. The direct antiglobulin test (DAT) detects antibodies or complement already bound to red blood cell surfaces in vivo. This test is critical for diagnosing autoimmune hemolytic anemia, investigating hemolytic transfusion reactions, and diagnosing hemolytic disease of the fetus and newborn. The indirect antiglobulin test (IAT) detects free antibodies circulating in serum or plasma. This test is essential for pre-transfusion antibody screening, crossmatching blood units for compatibility, and prenatal antibody screening in pregnant women. Polyspecific Coombs antisera (like the green-colored reagent shown) contains antibodies against both IgG and C3d complement, providing broad-spectrum detection. When the polyspecific reagent produces a positive result, monospecific antisera (anti-IgG alone or anti-C3d alone) are used for follow-up testing to characterize whether red cells are coated with IgG antibodies, complement, or both. This differentiation is clinically important because it helps determine the cause and clinical significance of the positive test. The reagent is typically dyed green using patent blue and tartrazine to allow easy visual identification during laboratory workflows where multiple reagents are used simultaneously. Storage at 2-8°C maintains reagent potency until the expiration date printed on the bottle, typically 18 to 24 months from manufacture. The reagent contains sodium azide (0.1% w/v) as a preservative, which inhibits bacterial growth but requires careful handling and disposal. Each dropper bottle delivers approximately 40 microliters per drop, allowing precise volumetric dosing during testing. The reagent must not be diluted and should not be used if turbid, as turbidity indicates bacterial contamination or protein aggregation that will compromise test performance. For distributors supplying blood banks, hospital transfusion services, reference immunohematology laboratories, and donor screening centers, Coombs antisera represents an essential reagent with consumption directly tied to transfusion volume and prenatal screening programs. Sara Wellness has been exporting immunohematology reagents and blood banking supplies from India for 15 years.

Acon Urine HCG Pregnancy Test Kit
Two lines or one. That's all a pregnancy test comes down to. But ask any pharmacist, clinic manager, or hospital procurement officer how much rides on those two little lines being reliably accurate — and the conversation gets more serious very quickly. Because a false result, in either direction, is not just a product failure. It's a human failure. The ACON urine hCG Pregnancy Test Kit is built on the understanding that reliability is non-negotiable. ACON Laboratories, headquartered in San Diego and one of the world's leading manufacturers of rapid diagnostic tests, uses a chromatographic immunoassay format that detects human chorionic gonadotropin — the pregnancy hormone — in urine at a sensitivity threshold of 25 mIU/mL. That's the level at which hCG is consistently detectable in clinically confirmed early pregnancies. The test is calibrated against the WHO International Standard (3rd edition) and has FDA clearance, two regulatory benchmarks that procurement managers across almost every market in the world recognise and accept as evidence of validated performance. The test works in three minutes. First morning urine is preferred for maximum accuracy, but the test can be performed at any point during the day. Each strip is individually foil-sealed, maintaining integrity until the moment of use. The result reads simply: two lines positive, one line negative, no line in the control window means the test is invalid and should be repeated. Nothing about the procedure requires training or lab equipment — which is precisely why this product moves so effectively across pharmacy chains, government health outreach programmes, antenatal clinics, and over-the-counter retail. For importers and distributors sourcing diagnostic rapid tests in volume, the ACON hCG strip kit offers the rare combination of brand recognition, regulatory compliance, and competitive unit economics. The 50-strip box format is the standard across institutional procurement — hospitals, clinics, and NGO supply chains all order in this configuration. Shelf life runs to 36 months from manufacture under standard storage between 2 and 30°C, which makes inventory planning straightforward even for markets with longer supply chains. Sara Wellness has been exporting diagnostic and lab supply products to buyers across Africa, the Middle East, and Southeast Asia for 15 years. Rapid test kits like the ACON hCG strip are among the highest-velocity SKUs in our catalogue. We understand volume pricing, documentation requirements for IVD products in different export markets, and the importance of consistent batch quality across repeated shipments. Reach out with your volume requirement and we will put together a quote worth looking at.

Albumin Reagent Kit (BCG Method)
Albumin is one of those analytes that tells a surprisingly large story from a very small sample. It is the most abundant protein in human plasma, making up roughly 60% of total serum protein, and its concentration reflects things as varied as liver function, nutritional status, kidney disease, and hydration. Low albumin can mean liver failure, malabsorption, or nephrotic syndrome. Elevated albumin can flag dehydration. It's one of the most frequently ordered biochemistry tests globally — which means the reagent that runs it needs to be reliable, reproducible, and available consistently. The Albumin Reagent Kit from Microsidd uses the Bromocresol Green (BCG) method, specifically the Doumas et al endpoint colorimetric approach. In plain terms: albumin in the sample binds to the BCG dye under pH conditions of 4.0 to 4.2, producing an albumin-BCG complex with a characteristic colour shift. The intensity of that colour, measured by the analyser at an absorbance peak of 625nm, is directly proportional to the albumin concentration in the sample. The kit is ready-to-use — no preparation steps, no mixing required before the assay. Results are generated in 3 minutes. The detection range runs from 0.130 g/dL to 7.5 g/dL, which covers everything from critically low albumin in acute liver failure to the upper limits of clinically relevant serum levels. The kit arrives as 2 x 50ml of liquid stable albumin reagent plus 1 albumin standard vial — everything needed to run and calibrate the assay. Reagents and the standard are both specially stabilized to ensure consistency across the kit's shelf life. The kit is compatible with semi-automated bench-top analysers and fully automated biochemistry analysers, making it usable across clinic labs, hospital biochemistry departments, and high-throughput reference laboratory settings. For laboratory reagent importers, medical distribution companies, and diagnostic supply chains across the Middle East, Africa, and Southeast Asia — albumin is a bread-and-butter biochemistry test. It is ordered alongside liver function panels, renal function panels, and nutritional assessments every day in every accredited laboratory. A reliable source for this reagent, priced competitively and backed by consistent supply, is worth more to a procurement manager than the occasional discount from an unreliable one. Sara Wellness exports laboratory reagents and biochemistry kits to buyers across multiple international markets. We understand cold-chain logistics, we understand CDSCO registration documentation, and we understand that laboratory procurement managers track expiry dates closely when evaluating suppliers. Every order we handle is accompanied by complete export documentation, and every cold-chain shipment leaves properly packaged for the journey. Fifteen years of this has taught us that the details matter more than the pitch. MOQs are flexible. Combined orders across multiple biochemistry reagent parameters move well and price well together.

Alere SD Bioline Dengue Duo Rapid Test Kit (NS1 Ag and IgG/IgM)
Dengue has a timing problem. Actually, dengue has several timing problems — but the most clinically inconvenient one is that the tests that work in the first few days of infection (antigen tests) stop being reliable around the same time that the tests for later-stage infection (antibody tests) start working. For years, this meant that a single test couldn't cover the full diagnostic window of a dengue case. Clinicians either over-tested, under-tested, or waited and watched while the patient got sicker. The SD Bioline Dengue Duo, developed by Standard Diagnostics (now part of Abbott / formerly Alere), was designed specifically to close that diagnostic gap. In one kit, two devices run simultaneously. The left device detects the NS1 antigen — a protein the dengue virus produces and releases into the bloodstream from day 1 of illness, making it detectable in the acute early phase when the patient first presents. The right device detects dengue-specific IgG and IgM antibodies, which appear later and persist through the convalescent phase. Together, the two devices cover all four dengue virus serotypes (DENV-1, 2, 3, and 4) and all clinically relevant stages from initial infection through recovery. Peer-reviewed studies published in the Journal of Biomedical Science and PMC (PubMed) confirm that combining NS1 with IgM and IgG results from this kit can bring overall diagnostic sensitivity to as high as 98.9% — a figure that single-parameter dengue tests simply cannot match. The NS1 device takes 100μL of serum, plasma, or whole blood. The IgG/IgM device takes just 10μL of sample plus 4 drops of the included assay diluent. Results on both devices are read at 15 to 20 minutes. Storage is ambient — 1 to 30°C — which removes cold-chain logistics from the equation entirely. That single feature makes this product far more practical for distribution into remote healthcare settings in tropical regions where dengue is endemic and cold storage infrastructure is unreliable. For diagnostic product importers and medical distributors sourcing for endemic markets — Southeast Asia, South Asia, sub-Saharan Africa, Latin America, and the Middle East — dengue diagnostics are a perennial, high-volume category. Dengue is not going away. Outbreaks continue to be declared across endemic regions every year, and the demand for reliable rapid tests spikes accordingly. The SD Bioline Dengue Duo carries CE marking and is distributed globally through Abbott's network, making it one of the most widely recognised and regulatory-accepted dengue rapid tests available. Sara Wellness supplies Abbott Bioline diagnostic kits to wholesale buyers across multiple international markets. No cold chain required means simpler logistics. Box-of-25 format moves efficiently at the institutional procurement level. Reach out with your volume requirements and target market — we will put a competitive quote together without unnecessary back-and-forth.
Alere Trueline HBsAg Rapid Test Kit
Hepatitis B is not dramatic in the way that some infectious diseases are. It doesn't announce itself with a distinctive rash or an unmistakable symptom. It moves quietly, often asymptomatically, establishing chronic infection in the majority of people who are exposed as infants and in a significant proportion of adults. Worldwide, approximately 292 million people are estimated to be living with chronic HBV infection — the majority of them in sub-Saharan Africa and Asia — and most of them don't know it yet. That is precisely why a fast, reliable, equipment-free HBsAg rapid test matters as much as it does. The Alere Trueline HBsAg Rapid Test Kit, manufactured by Alere Medical Pvt Ltd and distributed under the SD Bioline/Alere brand, is a lateral flow immunochromatographic assay designed to detect Hepatitis B surface antigen (HBsAg) in serum, plasma, or whole blood — from both venipuncture and fingerstick collection. The test requires 50μL of sample. Results appear in 15 minutes. Two red bars in the result window is positive. One bar is negative. No bar means the test is invalid. That's the entire user protocol. Sensitivity is confirmed at 95.16% and specificity at 99.95% from GloriaExports' cited performance data — performance independently validated in a multicentre prospective study published in the Journal of Medical Virology, which found whole blood sensitivity between 97.2 and 98.6% across sample collection methods, meeting both EU (0.13 IU/mL) and WHO (0.1 IU/mL) analytical sensitivity requirements. These are not numbers manufacturers generate alone. They are numbers from peer-reviewed clinical evaluation. Storage at 2 to 30°C means ambient room temperature throughout. No refrigerator. No cold chain logistics. No expensive packaging requirements for the last mile of distribution into peripheral health facilities. For healthcare system procurement across Africa, South Asia, and Southeast Asia — markets where HBV burden is highest and cold storage infrastructure is most uneven — that storage specification is not a convenience, it is a clinical programme enabler. For diagnostic product importers and medical distributors sourcing for endemic markets, HBsAg testing has sustained demand regardless of the health system level. Primary care clinics, antenatal screening programmes, blood banks, insurance pre-screening panels, and public health outreach camps all order this test. The Alere/Abbott brand name carries recognition across these procurement channels globally, which reduces the time needed to place a new product into a new market. Sara Wellness supplies Alere diagnostic kits to wholesale buyers across Africa, the Middle East, and Southeast Asia. We understand ambient storage shipping, we understand the documentation that rapid diagnostic test imports require across different regulatory markets, and we don't keep buyers waiting for answers. Fifteen years in this business means we've handled this product category more times than most of our competitors. Reach out with a volume requirement and a destination market, and we'll make the pricing worth your time.

Alere Trueline Malaria Rapid Test Kit (Ag Pf/Pan — HRP2 and pLDH)
There is a long-running debate in malaria diagnostics about whether the parasite count visible under a microscope is still the gold standard, or whether rapid antigen tests have effectively retired that argument in field settings. The answer, most health systems have quietly concluded, is: it depends on where the test is being done and by whom. In a well-staffed reference laboratory with experienced microscopists, microscopy wins on granularity. In every other setting — remote health posts, community screening camps, peripheral clinics, and anywhere that trained microscopists are not available at 2am — the rapid test is the practical standard of care. And it has been for some time. The Alere Trueline Malaria Rapid Test Kit (Ag Pf/Pan) is a one-step immunochromatographic assay that simultaneously detects two separate targets in a single test device. The first target is HRP-II (histidine-rich protein 2) — a protein produced specifically by Plasmodium falciparum, the most lethal malaria species. The second target is pLDH (pan-specific Plasmodium lactate dehydrogenase) — an enzyme produced by all four clinically significant Plasmodium species: P. falciparum, P. vivax, P. malariae, and P. ovale. The combination means the test not only confirms or excludes a malaria infection but differentiates a P. falciparum infection from a non-falciparum infection in a single cassette reading. That distinction matters clinically because P. falciparum carries the highest risk of severe and fatal disease and requires more urgent treatment. Performance figures, confirmed by Abbott Global Point of Care, place P.f HRP2 sensitivity at 99.7% and specificity at 99.5%. Pan pLDH sensitivity is 95.5% with specificity at 99.5%. These are the numbers published on the manufacturer's own product page — not estimates from a single study. The test uses 5μL of fingerstick whole blood. Results appear in approximately 20 minutes. Storage is 2 to 30°C throughout shelf life. No cold chain. No refrigerator. No generator-dependent equipment. For diagnostic distributors and healthcare importers supplying malaria-endemic markets across sub-Saharan Africa, South Asia, and Southeast Asia — the Alere/Abbott Bioline malaria test line is among the most WHO-evaluated and field-validated rapid diagnostic test brands available globally. Procurement managers in national malaria programmes and NGO supply chains recognise the product without explanation. The Trueline variant, manufactured by Alere Medical Pvt Ltd in India, offers the same dual-parameter Pf/Pan coverage with local regulatory compliance and export documentation for Indian-origin sourcing. Sara Wellness has supplied malaria diagnostic kits and other point-of-care tests to buyers across multiple international markets for 15 years. We handle all export documentation, understand in-country registration requirements in major markets, and keep communication direct and practical. Bulk orders, mixed SKU orders combining multiple rapid test parameters, and repeat supply agreements all welcome. The quote process is quick and the pricing is honest.
Alkaline Phosphatase Liquid Stable Reagent Kit
Clinical chemistry has a short list of tests that appear on almost every biochemistry panel, every day, in virtually every laboratory in the world. Alkaline phosphatase is on that list. It is not the most famous test — that distinction probably belongs to glucose or creatinine — but it is arguably one of the most quietly informative ones. When ALP rises, the laboratory report is beginning a story. Is it the liver? Is it bone? Is the biliary tract obstructed? Is this an osteoblastic process? The ALP result is rarely the end of a diagnostic conversation. More often, it's the thing that starts it. The Infinite brand Alkaline Phosphatase Liquid Stable Reagent Kit, manufactured and distributed by Accurex Biomedical Pvt Ltd, is the workhorse format for ALP testing in Indian clinical laboratory supply chains — and increasingly for export buyers sourcing Indian biochemistry reagents for their markets. The test uses the IFCC-optimised pNPP (p-nitrophenyl phosphate) kinetic method: ALP in the patient sample catalyses the hydrolysis of the pNPP substrate, converting it to p-nitrophenol, which produces a measurable colour change at 405nm. The rate of that colour change, measured by the analyser as ΔA per minute, is directly proportional to the ALP activity in the sample. The result is expressed in IU/L or U/L at 37°C. Both reagents — R1 and R2 — arrive ready to use. No reconstitution. No powder to weigh, no diluent to add, no intermediate preparation step before the assay. Liquid stability is the whole point of the product name, and it delivers: reagents are stable at 2–8°C until the expiry date printed on the label, and the working reagent once prepared is stable for 7 days under the same conditions. The kit is linear up to 2000 U/L, covering the full range from normal to severely elevated values without requiring dilution in the vast majority of samples. It is compatible with both semi-automated bench-top analysers and high-throughput fully automated biochemistry analysers. For laboratory reagent importers and clinical diagnostics distributors across the Middle East, Africa, and Southeast Asia — the ALP reagent is not a niche product. It is ordered every procurement cycle, because it runs every clinical chemistry panel. A supplier who can deliver this product consistently, on schedule, with the right documentation, earns a position in the supply chain that is difficult to displace. Sara Wellness exports Infinite brand biochemistry reagents and other Indian IVD products to wholesale buyers internationally. We understand cold-chain packaging, CDSCO registration documentation, and the batch-level expiry tracking that laboratory procurement teams require. Fifteen years of biochemistry reagent export experience means that the paperwork process is as organised as the logistics. MOQs are flexible, and combined orders across multiple biochemistry parameters move well and price competitively together.

Erba Uric Acid Biochemistry Reagent Kit (Uricase Method — Liquid Stable)
Gout has been called the disease of kings and the king of diseases. Both descriptions are accurate in their own way. For centuries it was associated with rich food and rich men — a reputation it earned honestly enough, given that uric acid accumulates when purines are metabolised and purines are plentiful in meat, shellfish, and alcohol. What has changed in recent decades is the clinical picture. Gout is no longer a condition of banquets. It is a condition of the modern metabolic environment — increasingly common, increasingly linked to hypertension, chronic kidney disease, and cardiovascular risk, and increasingly in front of general practitioners, nephrologists, and rheumatologists who need a reliable, consistent uric acid result to make treatment decisions. The Erba Mannheim Uric Acid Biochemistry Reagent Kit is a single-reagent, liquid stable enzymatic assay for the quantitative in vitro determination of uric acid in human serum, plasma, and urine. Erba Diagnostics Mannheim is a German in vitro diagnostics brand with a significant installed base across European and emerging market clinical laboratories, particularly through the Erba Chem semi-automated analyser range that dominates mid-tier hospital laboratory settings across Africa, the Middle East, and South Asia. The chemistry is the Uricase-Trinder / PAP endpoint method. Uricase converts uric acid to allantoin and hydrogen peroxide. The hydrogen peroxide then reacts with 4-aminoantipyrine and DHBS in the presence of peroxidase, producing a red quinoneimine dye. Absorbance of the coloured product is measured at 510nm and is directly proportional to the uric acid concentration. The reaction is complete, specific, and well-characterised — the same chemistry used by most major clinical chemistry platforms globally for serum uric acid quantification. Both pack sizes — 2x50ml (FBCEM0068) and 4x50ml (FBCEM0069) — arrive as a single ready-to-use liquid reagent with no mixing or reconstitution required. On-board stability is a minimum of 30 days at 2–10°C once loaded on the analyser, which is commercially relevant for high-throughput automated analysers running continuous workflows. Linearity extends to 25 mg/dL, covering both normal and severely elevated values including those seen in acute gout flares and tophaceous gout. Known interferents — haemoglobin up to 10 g/L, bilirubin up to 40 mg/dL, and triglycerides up to 2000 mg/dL — do not affect the assay at these concentrations. For laboratory reagent importers and clinical chemistry distributors supplying markets where Erba Chem analysers are widely installed, the Erba reagent line is the natural sourcing choice. Running the instrument's own brand reagents on the Erba Chem platform avoids the compatibility validation burden and is often the procurement default for hospital lab managers who have standardised on this analyser. Sara Wellness exports Erba Mannheim reagents and other Indian-origin and imported clinical chemistry products to wholesale buyers across Africa, the Middle East, and Southeast Asia. We know cold-chain packaging, we know what export documentation your regulatory authority requires, and we have been doing this long enough that the process runs without surprises. Reach out with your volume requirement and we will respond with a real number.

Am I Rapid hCG Pregnancy Test Device (Biotron — WHO-GMP)
The name is the whole point. Am I? Two syllables. The question that about 20 million women globally ask every month. The product doesn't dance around what it is or what it does — and neither should its description. The am i Rapid hCG Device, manufactured by Biotron to WHO-GMP standards, is a one-step lateral flow chromatographic immunoassay for the qualitative detection of human chorionic gonadotropin (hCG) in urine. hCG is the hormone produced by the developing placenta beginning shortly after fertilisation. It appears in urine as early as 7 to 10 days post-conception and rises rapidly through the first trimester, reaching concentrations well above the test's detection threshold of 25 mIU/mL by the day of the first missed period. That 25 mIU/mL sensitivity threshold means the test catches pregnancies at the earliest clinically relevant point — not a week after the missed period, not at a point when the information is already obvious. At the moment when the question first forms and actually needs an answer. The device format — a cassette rather than a strip — is the clinical and procurement standard for institutional use. Dispensing pipettes are included per device. The user adds 3 to 4 drops of urine to the sample well and reads the result at 3 to 5 minutes. Two lines is positive. One line is negative. No control line means the test is invalid. Specificity exceeds 99%, calibrated against the WHO International Standard for hCG. Known cross-reactants — LH at 300 mIU/mL, FSH at 1,000 mIU/mL, TSH at 1,000 μIU/mL — do not interfere with the assay at these concentrations. Storage is ambient at 2 to 30°C throughout the 24-month shelf life. No refrigeration. No cold chain logistics. No special handling between manufacture and end use. For diagnostic product importers and medical distributors supplying pharmacy chains, antenatal clinics, government maternal health programmes, and hospital outpatient departments — the hCG rapid test device is among the highest-volume rapid test SKUs in any reproductive health supply chain. It is ordered in every procurement cycle without exception, across every market where antenatal care or family planning services operate. The WHO-GMP certification on the am i Rapid device signals a manufacturing standard that satisfies procurement requirements in regulated import markets across the Middle East, Africa, and South and Southeast Asia. Sara Wellness exports Biotron and other WHO-GMP certified rapid diagnostic devices to wholesale buyers across multiple international markets. We know the reproductive health supply chain — the volume patterns, the procurement calendar, and the documentation that import authorities require for IVD devices in different markets. Fifteen years in this category means we have handled this SKU more times than it takes to get very good at it. Send us your volume requirement and your destination, and we'll have a quote ready in 24 hours.

Amylase Reagent Kit Liquid Stable
Pancreatitis has a particular skill for arriving without much warning and demanding immediate answers. A patient presents with sudden severe abdominal pain radiating to the back, nausea, and a clinical picture that could fit half a dozen diagnoses. The test that most quickly and reliably points toward the pancreas as the source is a serum amylase. It rises within 2 to 12 hours of acute pancreatitis onset and remains elevated for 3 to 7 days — a time window that makes it useful in both emergency presentation and follow-up. For emergency departments, general medicine wards, and any laboratory running clinical chemistry panels on acutely unwell patients, amylase is not an esoteric test. It is a core panel component. The Erba Mannheim Amylase Reagent Kit Liquid Stable uses the CNPG3 (2-chloro-4-nitrophenyl-α-D-maltotrioside) kinetic method. Amylase in the patient's sample cleaves the CNPG3 substrate, releasing 2-chloro-4-nitrophenol (CNP). The rate of CNP formation — measured as ΔA per minute at 405nm — is directly proportional to the amylase activity in the sample. The result is expressed in U/L or IU/L at 37°C. This is the IFCC-recommended substrate for clinical amylase measurement, making the method internationally standardised and comparable across different laboratories and platforms. The reagent is ready to use. No reconstitution. No preparation steps. It arrives as a liquid stable single-reagent format and goes directly from the bottle into the analyser. Lower detection limit is 3 U/L; linearity extends to 2000 U/L without dilution. Samples above this threshold require dilution with normal saline (9 g/L) and result multiplication by 5 — which is a clear, documented protocol, not an improvised workaround. The kit is compatible with both semi-automated bench-top analysers and Erba Mannheim's own automated XL analyser series through the dedicated SysPacks configuration. For laboratory reagent importers and clinical chemistry distributors — particularly those supplying markets where the Erba Chem and Erba XL analysers have significant installed base — the amylase kit is a repeat-order item on every procurement cycle. Emergency department workload drives daily amylase testing. Hospital labs cannot substitute or defer this test without clinical risk to patients. That makes it as close to a guaranteed volume commitment as exists in the reagent supply business. Erba Mannheim is a German diagnostic brand with genuine market penetration across Africa, the Middle East, and South Asia, particularly through the mid-tier hospital laboratory segment. Supplying Erba-brand reagents to Erba-analyser customers is the most friction-free procurement conversation there is — the customer already knows the brand, already runs the instrument, and already knows what the reagent should do. Sara Wellness exports Erba Mannheim reagents and other clinical chemistry kits to wholesale buyers across multiple international markets. We handle cold-chain packaging, export documentation, and batch-level traceability records without needing to be asked. Fifteen years of biochemistry reagent export gives us a practical understanding of what lab procurement managers require at every step. Combined orders across multiple Erba Mannheim parameters move well and price accordingly.
Amylase Reagent 12ml Liquid Stable
There is a test that emergency doctors order almost reflexively when a patient walks in with severe upper abdominal pain. It comes before the ultrasound booking, before the surgical consult, sometimes before the full history is complete. Serum amylase. Four words. One tube of blood. A number that, within 30 minutes of the sample reaching the laboratory, can begin to confirm or rule out one of the most common acute abdominal emergencies managed in hospital medicine globally. The Infinite Amylase Reagent 12ml Liquid Stable, produced by Accurex Biomedical — India's first biochemical reagent manufacturing company — is the small-volume format designed for clinical laboratories running amylase on semi-automated bench-top analysers. The 12ml format is specifically practical in settings where a single reagent refill covers a defined daily or weekly assay run without opening large-volume bottles that reduce in activity with repeated exposure. Clinical chemistry laboratories in mid-sized hospitals, district hospitals, and stand-alone diagnostic centres across export markets are the natural home for this format. The chemistry is the CNPG3 substrate method — the IFCC-recommended kinetic approach for alpha-amylase measurement. Amylase in the patient's sample cleaves the synthetic substrate, producing a chromogenic product whose rate of formation is measured at 405nm and expressed in U/L at 37°C. The assay is specific, well-characterised, and produces results that are directly comparable across laboratories using the same IFCC-standardised substrate chemistry. The reagent is liquid stable — ready to use, no reconstitution, no weighing, no preparation. It goes from the storage refrigerator into the analyser's reagent position and runs. Pack size matters more than it is sometimes given credit for in clinical laboratory procurement. A 12ml format — rather than a large-volume bottle — reduces the risk of on-analyser reagent degradation, which is a real and documented source of quality issues in laboratories where daily test volumes are modest and reagent bottles are open for extended periods between runs. Ordering the right volume format for the actual throughput of the analyser is a decision that affects result quality at the assay level, not just cost per test at the procurement level. For laboratory reagent importers and clinical chemistry distributors building out portfolios for mid-tier hospital and diagnostic laboratory markets — Infinite is an Accurex brand with 60-plus years of Indian biochemistry reagent manufacturing behind it. It is not a new entrant to the market. It is recognised across South Asia, parts of the Middle East, and several African markets as a cost-competitive, clinically validated alternative to premium European reagent brands. Sara Wellness exports Infinite brand reagents and other Accurex Biomedical products to wholesale buyers across multiple international markets. We know cold-chain documentation, we know batch-level expiry records, and we know that reliable delivery on repeat orders matters more than the first-order pitch. MOQs are flexible. Combined orders across multiple Infinite biochemistry parameters get the best pricing. We are easy to talk to and even easier to order from.

Anti D IgG Monoclonal Blood Grouping Reagent
The D antigen is not the only antigen in the Rh blood group system — there are over 50 antigens in the system — but it is by far the most clinically significant. An Rh-negative patient who receives Rh-positive blood and makes Anti-D antibodies has a complication that is now manageable. An Rh-negative mother whose Rh-positive fetus bleeds into her circulation, and who is not given Rh immunoglobulin prophylaxis, risks sensitisation that could cause haemolytic disease of the newborn in a subsequent pregnancy. The clinical stakes around D antigen determination have been understood for 75 years and the testing procedure remains unchanged: test the red cells with Anti-D and read the agglutination. The Anti-D IgG Monoclonal Blood Grouping Reagent is a class IgG antibody — which makes it distinctly different from the IgM Anti-D used in routine forward slide grouping. IgG antibodies do not agglutinate red cells in saline directly. Instead, the IgG Anti-D sensitises D antigen-positive red cells, and agglutination is then detected in the antiglobulin (Coombs) phase of testing. This makes the IgG format specifically useful for detecting the D antigen through the indirect antiglobulin test (IAT) and for Du (weak D) testing — the additional step required when routine direct agglutination with IgM Anti-D gives a negative result but D-negativity must be definitively confirmed, particularly for blood donors. The clinical scenario where IgG Anti-D is most often specifically required is weak D testing. Blood donors who test negative by routine IgM Anti-D reagent must have their red cells further tested for the Du antigen — a weaker expression of the D antigen that, if present on donor blood labelled as Rh-negative, could sensitise an Rh-negative recipient. The IgG Anti-D used in the indirect antiglobulin test is the reagent for this confirmation step. Available in a 10ml dropper bottle for standard laboratory use and a 1L bulk format for high-throughput reference laboratories and blood centres, the Anti-D IgG Monoclonal Reagent is validated for slide and tube techniques and is designed for use by operators trained in serological blood banking procedures. Sara Wellness exports blood grouping reagents including Anti-D IgG format to wholesale buyers across Africa, the Middle East, and Southeast Asia. Blood bank reagent supply chains require documented cold-chain handling, and we have the export experience to support that requirement end to end. Orders can be combined across the full blood grouping reagent range — Anti-A, Anti-B, Anti-D IgM, Anti-D IgG, and Anti-AB — for consolidated shipping and pricing efficiency.

Anti A1 Lectin Blood Grouping Reagent (Dolichos biflorus)
Not all blood grouping tests are created equal, and not all blood grouping questions are as simple as A, B, or O. Every so often, a patient with blood group A comes back with a result that does not quite fit the expected serology. The forward group says A. The reverse group says something slightly different. And the blood banker, instinctively, reaches for the Anti-A1 Lectin. Anti-A1 Lectin is the specialist reagent within the ABO subgrouping toolkit. It is derived from an extract of Dolichos biflorus seeds — the same plant extract that blood bankers have relied on to differentiate the A1 subgroup from A2 and weaker A subgroups for over seven decades. In a population of group A individuals, approximately 80% are A1 and approximately 20% are A2 or weaker. The distinction matters clinically: some A2 and A2B individuals make an alloanti-A1 antibody that behaves as an unexpected serum antibody in ABO reverse grouping, producing the discrepancy that sent the blood banker searching for this reagent in the first place. The test is straightforward. One drop of Anti-A1 Lectin is mixed with a drop of the patient's red cell suspension on a slide, or added to a tube for centrifugation. Red cells that carry the A1 antigen will agglutinate. Those that are A2 or weaker will not. The result, read within one minute on a slide or after centrifugation in a tube, resolves the discrepancy and classifies the patient definitively. The reagent is prepared as a stabilised aqueous extract of Dolichos biflorus seeds, diluted in phosphate-buffered saline containing bovine serum albumin. It is supplied ready to use with no further dilution required, and contains 0.1% sodium azide as a preservative. For blood banks that manage complex serology workups, for regional reference laboratories, and for hospital blood banking services that encounter A subgroup discrepancies, this reagent is a non-negotiable part of the immunohaematology reagent cabinet. It does not move in the same volumes as Anti-A and Anti-B, but when it is needed, nothing else substitutes for it. Anti-A1 Lectin is available in 5ml and 10ml dropper vials for routine laboratory use, and in a 1L bulk format for reference laboratories or high-volume procurement. Sara Wellness exports blood grouping reagents and immunohaematology supplies to wholesale buyers and IVD distributors across Africa, the Middle East, and Southeast Asia. We understand cold-chain export requirements and import documentation for regulated markets. Fifteen years of IVD export experience mean these shipments run correctly. Combined orders across the full blood grouping reagent range receive the best pricing.

Anti A Monoclonal Blood Grouping Reagent (IgM)
Blood typing is one of the oldest and most consequential diagnostic procedures in clinical medicine. Get it right and a transfusion saves a life. Get it wrong and the consequences are irreversible. The Anti-A Monoclonal Blood Grouping Reagent is the first of the three essential reagents that every blood bank, transfusion centre, and pre-operative blood screening laboratory keeps permanently in stock — alongside Anti-B and Anti-D — because without these three, nothing in blood banking moves. The Anti-A Monoclonal Reagent is a murine monoclonal IgM antibody produced in vitro from a selected hybridoma cell line derived by fusion of mouse antibody-producing B-lymphocytes with mouse myeloma cells. Because it originates from a single cell line, every molecule of antibody in the bottle is structurally identical with the same specificity, the same avidity, and the same agglutination characteristics. That batch-to-batch consistency is precisely what makes monoclonal reagents the global replacement for polyclonal antisera: there is no variation from one production lot to the next and no dependence on human donor serum. When a drop of this reagent is mixed with a drop of the patient's washed red cell suspension on a slide or in a tube, it will agglutinate red cells carrying the A antigen. Strong, visible clumping within 15 to 30 seconds on a slide is a positive result. No agglutination is negative. The technique is simple enough for a junior blood bank technician to perform correctly, which is exactly why it has remained the standard forward grouping method in settings ranging from fully equipped reference laboratories to district hospital blood banks. The reagent is internationally colour-coded blue — per the global convention that makes Anti-A bottles universally distinguishable from Anti-B (yellow) and Anti-D regardless of language or label design. It is supplied in a calibrated dropper bottle for consistent drop-to-drop volume in slide and tube tests. Titre is at minimum 1:256 for Anti-A as per international quality standards. Shelf life is 24 to 36 months from manufacture when stored at 2 to 8°C. The 10ml bottle is the standard institutional format; 5ml, 6x10ml box, and 1L bulk sizes are available for different throughput requirements. For laboratory reagent importers and blood bank supply distributors — blood grouping reagents have non-negotiable reorder cycles. Every blood bank runs these reagents every day. Stock-outs are not acceptable. The supply chain for Anti-A must be predictable, consistent, and well-documented. Sara Wellness exports monoclonal blood grouping reagents and other IVD products to wholesale buyers across Africa, the Middle East, and Southeast Asia. We understand cold-chain requirements, CDSCO documentation, and batch-level certificate requirements for import clearance. Fifteen years of IVD reagent export experience means the process runs predictably. Contact us with your volume requirement and we will respond promptly.

Anti B Monoclonal Blood Grouping Reagent (IgM)
In a blood bank, the Anti-B reagent does not get more attention than Anti-A. They are ordered together, used together, and interpreted together. Blood group B is present in roughly 8 to 11 percent of most populations depending on ethnicity — and in some South Asian and African populations, the frequency is considerably higher. Every single one of those patients needs their B antigen detected correctly before they can receive a blood transfusion. Which means Anti-B is not optional, it is not substitutable, and it is never allowed to run out. The Anti-B Monoclonal Blood Grouping Reagent is a murine monoclonal IgM antibody produced from a single hybridoma cell line by in vitro culture. The antibody is specific for the B antigen on human red blood cells. It will produce visible agglutination of B antigen-carrying red cells in slide, tube, or microplate formats within seconds. No agglutination means no B antigen — the patient is group A, group O, or requires further investigation. The result is read macroscopically. No equipment is required beyond a clean surface and good lighting. The logic of using monoclonal antibodies for blood grouping is now well-established. Human donor serum — the polyclonal source that blood grouping antisera relied on for decades — varies between donors, varies between bleeds from the same donor, and introduces the spectre of additional unexpected antibodies in the reagent. A monoclonal hybridoma cell line that has been selected and validated produces the same antibody at the same concentration with the same specificity every time. The ABO grouping of every patient processed over every year of the reagent's shelf life will be performed with functionally identical reagent. The bottle is colour-coded YELLOW — the international standard for Anti-B blood grouping reagents, established to prevent mix-up with Anti-A (blue) and Anti-D (clear or grey depending on the manufacturer's convention). The calibrated dropper ensures the correct volume is dispensed per test without measuring. Titre is at minimum 1:256. Shelf life is 24 to 36 months stored at 2 to 8°C. For blood bank supply distributors and laboratory reagent importers — Anti-A and Anti-B are almost always ordered together because they are used together. Every forward grouping procedure needs both. Procurement managers who understand this buy both in the same order, from the same supplier, to keep the supply chain simple. Sara Wellness supplies both, and the rest of the blood grouping reagent range, to wholesale buyers across multiple international markets. Fifteen years of IVD export experience means the documentation for regulated markets is handled correctly. Combined orders across the ABO-D reagent range price competitively.

Anti D Monoclonal Antibody Reagent (IgM+IgG Blend — Rh Typing)
If there is one test result in blood banking that cannot wait, cannot be approximate, and cannot be re-done after a transfusion has been given — it is the Rh(D) type. Getting this result wrong is not a minor analytical error. It is the kind of error that has life-and-death consequences across both transfusion medicine and obstetrics. The history of Rh incompatibility in medicine, and the decades of work that went into developing reliable Anti-D reagents, is a story about what happens when this result goes wrong and what it takes to make sure it does not. The Anti-D Monoclonal Antibody Reagent is the IgM+IgG blend format — the most complete Anti-D reagent configuration for routine clinical use. The IgM component provides the direct saline agglutination required for rapid slide and tube testing in routine Rh typing. The IgG component provides the sensitivity for detecting weak D-expressing red cells in the antiglobulin phase. Used together as a blend, the reagent delivers both the speed of a direct slide test and the sensitivity of the Coombs phase without requiring separate sequential testing, unless protocol dictates otherwise. The blend is produced from human and murine hybridoma cell lines — multiple clones that together cover the full spectrum of D antigen expression, including partial D variants and category DVI cells. The reagent will cause direct agglutination of D antigen-positive red cells that carry the full D antigen, and indirect agglutination of category DVI cells in the antiglobulin phase. Red cells that show no reaction in either phase are classified as D-negative, subject to the laboratory's Du testing protocol for blood donors. The titre is at minimum 1:128 for Anti-D as per the Tulip Eryscreen combipack confirmed specification — lower than Anti-A and Anti-B (which run at 1:256) because Anti-D detection is intrinsically more demanding and this titre represents the validated clinical performance threshold. The reagent is supplied in a calibrated dropper bottle for consistent drop volume in testing. It is available individually in 5ml and 10ml bottles, in a 1L bulk format for high-throughput blood centres, and as part of the Tulip Eryscreen 3x10ml combipack (Anti-A + Anti-B + Anti-D) — the single-box solution for laboratories that prefer to source all three ABO-D grouping reagents from one supplier and one lot. For blood bank supply distributors and diagnostic reagent importers, Anti-D is the most safety-critical of the three ABO-D grouping reagents and the one for which procurement reliability matters most. A supply interruption in Anti-D stops blood typing. Sara Wellness supplies Anti-D monoclonal blend reagent in the individual bottle and combipack formats, exports with full cold-chain documentation, and maintains adequate stock for consistent repeat supply. Fifteen years in the IVD export business means these supply chains run as they should.

Blood Grouping Kit (ABO & Rh Typing — Anti-ABD Combipack)
Three reagents. Three dropper bottles. One box. And with those three components, a laboratory can determine the blood group and Rh type of every patient who walks through the door — before any transfusion, before any surgical procedure, and before any obstetric decision that depends on knowing whether a mother is Rh-negative. The Blood Grouping Combipack — Anti-A, Anti-B, and Anti-D together in a single packaged kit — exists because these three reagents are always used together in ABO-D blood grouping. No blood bank runs Anti-A without Anti-B. No transfusion centre does Rh typing without the other two. They are ordered together, stored together, and expire together. Packaging them as a combipack simply recognises the clinical reality that they are a three-part test system, not three independent products. The kit contains monoclonal IgM antibodies for Anti-A (colour-coded blue, titre minimum 1:256), Anti-B (colour-coded yellow, titre minimum 1:256), and Anti-D (titre minimum 1:128). All three are ready to use directly from the dropper bottles with no dilution. The blood grouping procedure is the most fundamental test in transfusion medicine and takes less than two minutes by the slide method: one drop of each reagent plus one drop of the patient's red cell suspension, mixed on a clean glass slide, read for agglutination at one minute. The result determines the patient's ABO group and Rh type definitively. For blood bank supply distributors, hospital procurement managers, and IVD product importers, blood grouping kits represent one of the most predictable procurement categories in the entire laboratory supply portfolio. Every blood group that is tested requires all three reagents. Consumption is directly proportional to patient throughput, and patient throughput does not go down. The kit format simplifies procurement by reducing the blood grouping reagent purchase to a single line item with a matched lot number and a single expiry date across all three bottles. The combipack is available in 3x5ml and 3x10ml formats for different laboratory throughput requirements. The 3x10ml format is the standard institutional size for hospital blood banks and transfusion centres. The 3x5ml format suits smaller clinics and district hospitals. Sara Wellness exports blood grouping kits and other IVD laboratory reagents to wholesale buyers across multiple international markets. Fifteen years of IVD export experience means the cold-chain documentation and import compliance requirements are handled correctly.

Turbilatex CRP Reagent Kit (Latex Turbidimetric — C-Reactive Protein)
C-reactive protein is one of those biomarkers that quietly does an outsized amount of work in the clinical laboratory. It shows up in the differential workup for bacterial infection, in the monitoring of inflammatory disease, in the post-surgical review of recovery progress, and in the risk stratification of cardiac patients. The test that measures it needs to be fast, quantitative, precise, and compatible with the automated biochemistry analysers that most clinical labs run. Turbilatex CRP checks every one of those boxes. The Turbilatex CRP Reagent Kit is a quantitative latex-enhanced immunoturbidimetric assay for the measurement of C-reactive protein in human serum or plasma. The principle is straightforward: anti-human CRP antibodies are adsorbed onto latex particles. When mixed with a patient sample containing CRP, the latex particles agglutinate in proportion to the CRP concentration. This agglutination produces an increase in absorbance that the analyser measures photometrically. The CRP concentration in the sample is then calculated by interpolation against a calibration curve. The reagent system is a two-component liquid format: R1 is a Tris buffer diluent, and R2 contains the anti-CRP antibody-coated latex particles. Both are ready to use without reconstitution. The assay is linear from a lower detection limit of 2 mg/L up to 150 mg/L, with samples exceeding the upper linearity requiring simple dilution with normal saline and reassay. Normal reference values are up to 6 mg/L, with laboratory-specific reference ranges recommended. The reagents remain stable on-board for a minimum of 30 days at 2-8°C once opened. The kit is compatible with a wide range of analysers — spectrophotometers, discrete semi-automated platforms, and fully automated biochemistry analysers. Manufacturers including Accurex (Infinite), Anamol, and others supply this kit format for use on platforms such as the B Auto 400, Unicorn 480, Bonavera Chem 400, Beaconic B400, Beaconic B200, and Beaconic Chem 400, among others. For clinical laboratory reagent distributors and IVD importers building biochemistry reagent portfolios — CRP is one of the highest-volume immunoturbidimetric tests in routine pathology. It is ordered across inpatient and outpatient settings with the same frequency as a liver function panel, and the consumption rate in busy laboratories is substantial. Sara Wellness exports clinical laboratory reagents and IVD diagnostic kits to wholesale buyers. Multiple pack formats are available. Cold-chain export and import documentation are handled correctly. Fifteen years of IVD export experience backs every shipment.

ATPL Urine Pot (Disposable Specimen Collection Container)
The urine specimen container is the product that every hospital uses in genuinely enormous quantities and almost nobody ever thinks carefully about until there is a problem with one. The cap leaks in transit. The lid is too tight to open with gloved hands. The graduation markings rub off before the sample reaches the laboratory. These are small frustrations that compound quickly when you are dealing with hundreds of samples per day across a pathology department. The ATPL Urine Pot is a disposable specimen collection container manufactured from medical-grade polypropylene. Polypropylene is the right material for this application — it is chemically inert (does not react with urine or any urinalysis reagents), transparent enough for direct visual inspection of sample volume and colour, and compatible with the full temperature range of laboratory handling including refrigeration. The wide-mouth design allows comfortable collection without patient difficulty, and the screw cap provides a reliable leak-proof seal for sample transport within the hospital or to external reference laboratories. The container is available in 60ml, 80ml, and 100ml volumes — the 60ml and 80ml sizes suited to routine urinalysis and dipstick testing where a moderate sample volume is sufficient, and the 100ml size providing extra capacity for patients who require a larger collection or for samples requiring multiple tests from a single collection. Moulded-in graduation markings on the container wall allow direct volume reading without measuring cylinders or additional equipment. The non-sterile variant is appropriate for the majority of urine tests: routine urinalysis, urine microscopy, pregnancy tests, drug screening, and biochemical urine tests. Sterile variants in individually wrapped packaging are available for urine culture and sensitivity (MC&S) testing, where sample contamination would invalidate the microbiological result. For hospital supply distributors, pathology laboratory consumable buyers, and clinical supply procurement officers — urine containers are a core, non-negotiable, high-velocity line item. The consumption volume across an active pathology department is predictable and continuous. Sara Wellness exports ATPL medical consumables to wholesale buyers. Volume pricing applies on bulk orders. MOQs are flexible for both trial and established supply contracts.

BD Vacutainer Blood Collection Tube (Plain Red — Serum)
There is an argument to be made that the BD Vacutainer blood collection tube is the most consequential piece of laboratory plasticware ever manufactured. That sounds like hyperbole until you calculate the number of diagnostic test results — and the number of clinical decisions — that depend on correctly collected, correctly clotted, and correctly processed blood samples every single day across the world's hospital laboratories. The Vacutainer system standardised how that collection happens, and the plain red serum tube is where a remarkable proportion of biochemistry and serology testing begins. The BD Vacutainer plain serum tube (red top) is a single-use, evacuated blood collection tube with no anticoagulant additive. Blood collected into this tube is allowed to clot completely — typically 30 to 60 minutes at room temperature — before centrifugation separates the clotted cellular fraction from serum. The resulting serum is then used for the widest range of clinical chemistry tests: liver function panel, kidney function panel, thyroid function tests, lipid profile, tumour markers, therapeutic drug monitoring, and most serology and immunoassay tests. Serum is the appropriate sample matrix for these tests because anticoagulants in tubes like EDTA or lithium heparin can interfere with the enzymatic or immunometric measurement methods. The tube is made from PET (polyethylene terephthalate) plastic — a material selected by BD specifically because it combines mechanical strength (very low breakage risk compared to glass), chemical inertness, and optical clarity for visual inspection of the sample. The interior is sterilised by gamma radiation. Tubes are manufactured under a carefully calibrated negative pressure so that exactly the correct nominal volume fills from the patient's vein without the operator controlling draw time — the tube simply fills to the calibrated volume and stops. For laboratory supply distributors supplying hospital pathology departments, commercial diagnostic laboratories, and clinical research organisations — BD Vacutainer serum tubes are among the highest-volume consumable lines in the entire laboratory supply category. Consumption is directly proportional to clinical activity and does not decline. Sara Wellness exports BD Vacutainer blood collection tubes and related phlebotomy consumables to wholesale buyers. Standard box of 100 and case quantities available across the full Vacutainer range.

BD Vacutainer EDTA Blood Collection Tube (Lavender/Purple — K2EDTA)
The haematology department runs on the lavender top tube. Every single day, in every hospital with a blood count analyser, the majority of the blood samples arriving in the laboratory are in lavender-capped EDTA tubes. CBCs. Differentials. Platelet counts. HbA1c. Blood films. ESR. Malaria antigen tests. Reticulocyte counts. Haemoglobinopathy screening. The entire field of haematology — the one that catches anaemia, leukaemia, thrombocytopenia, and a host of other conditions in their earliest detectable stages — depends on a tube that costs almost nothing and works every time. The BD Vacutainer EDTA Blood Collection Tube (lavender or purple top) is an evacuated plastic blood collection tube containing K2EDTA (dipotassium ethylenediaminetetraacetic acid) as the anticoagulant additive. EDTA is the anticoagulant of choice for haematological testing because it preserves cell morphology and prevents clotting without significantly affecting red blood cell volume or white blood cell differential counts — unlike heparin which can cause leukocyte clumping and is unsuitable for differential counting. K2EDTA is supplied as a dry spray-coat on the interior tube walls at a precise concentration — 1.8 mg EDTA per millilitre of blood (ISO 6710 standard concentration). When blood fills the tube and the tube is inverted gently 8 to 10 times to dissolve the EDTA and mix it uniformly with the blood sample, the calcium ions in the blood are chelated by the EDTA, preventing the calcium-dependent clotting cascade from proceeding. The result is a fully anticoagulated whole blood sample that can be analysed on any haematology analyser without clotting artefact. The BD Vacutainer EDTA tube uses PET plastic construction with a BD Hemogard lavender closure. The Hemogard design eliminates direct skin contact with blood on the stopper surface when the tube is opened, a feature developed to reduce occupational exposure in the laboratory. Tubes are sterilised by gamma radiation and supplied at calibrated draw volumes from 2ml (paediatric) to 10ml (adult large-volume). For laboratory supply distributors and hospital procurement managers — lavender EDTA tubes are the highest-volume blood collection tube SKU in virtually every hospital laboratory supply budget. The CBC is the most ordered single blood test in clinical medicine globally. Sara Wellness exports the full BD Vacutainer range to wholesale buyers, with EDTA tubes available in standard box and bulk case quantities.

BD Vacutainer SST Advance Blood Collection Tube (Gold — Serum Separator)
Let us say the morning phlebotomy run is done. Forty-seven patients. A mix of inpatient wards and outpatient clinics. The tubes are in the centrifuge. Now here is the part that most people do not think about: after centrifugation, someone has to separate the serum from the clot pellet cleanly enough that the sample can be poured or pipetted into the analyser cup without aspirating any cellular material. With a plain red tube, that means careful pouring, or aliquoting, or gel-free laboratory technique. With the SST tube, the gel does that work automatically. The BD Vacutainer SST Advance tube is a gold-top serum separator tube. SST stands for Serum Separator Tube. The gold colour specifically denotes the gold BD Hemogard closure — a push-pull plastic closure that, unlike traditional rubber stoppers, prevents direct contact between the healthcare worker's fingers and the blood on the stopper surface when the tube is opened. The Hemogard closure also reduces the risk of blood splatter during opening, a meaningful occupational safety feature when processing high-throughput sample batches. The tube's interior has two critical features. First, the walls are spray-coated with micronized silica particles — these accelerate the natural blood clotting process so that a robust, complete clot forms in approximately 30 minutes at room temperature. This is faster and more reliable than waiting for spontaneous clotting in a plain tube. Second, and the SST's defining feature, a thixotropic polymer gel separator sits at the bottom of the tube before centrifugation. The gel has a specific gravity between that of serum and blood cells. During centrifugation (1000-1300 RCF for 10 minutes), the gel migrates upward through the cellular pellet and settles at the serum-cell interface, forming a stable physical barrier between the serum above and the clot below. The serum can then be poured or aspirated directly from above the gel barrier without cellular contamination, without the need for aliquoting into a separate tube. The SST is the workhorse serum tube for routine clinical chemistry: liver function, kidney function, lipids, glucose, thyroid, hormones, cardiac markers, vitamin D — virtually every standard biochemistry panel that a clinical laboratory processes in volume. For laboratory supply distributors — SST gold tubes consistently rank as the highest-volume serum tube SKU in hospital laboratory procurement. Sara Wellness exports the full BD Vacutainer SST range to wholesale buyers, with gold-top SST tubes available in the complete volume range and in box and case quantities.
BD Vacutainer Citrate Tube (Light Blue — Sodium Citrate, Coagulation Testing)
Coagulation testing is the one area of laboratory medicine where a pre-analytical error can be catastrophic rather than just inconvenient. A PT/INR drawn in the wrong tube — a plain red tube, say, because someone reached for the wrong colour — cannot be corrected by rerunning the analysis. The clotting factors have already interacted. The sample is invalid. For a patient on warfarin being managed at a therapeutic INR, that wrong tube can mean a missed dose adjustment, a bleed, or an unnecessary anticoagulation reversal. The blue top matters. The BD Vacutainer Citrate Tube is the global reference standard for coagulation blood collection. The light blue BD Hemogard closure has meant coagulation tube for more than 75 years of laboratory practice. The additive inside is 3.2% buffered sodium citrate (0.109M) — a calcium chelator that arrests the coagulation cascade at the moment of collection by binding the calcium ions required for Factor activation. This preserves the patient's clotting factor levels in the same state as when the blood left the vein, allowing the laboratory's coagulation analyser to run PT, APTT, fibrinogen, D-dimer, and coagulation factor assays on a sample that accurately reflects in vivo clotting status. The blood-to-citrate ratio of 9:1 is not a guideline — it is a hard requirement. Under-filling the tube changes this ratio, dilutes the clotting factors relative to the citrate concentration, and produces falsely prolonged clotting times. Over-filling reduces the citrate-to-calcium ratio and may allow partial activation. BD addresses this with a 360° etched fill line on the tube body that marks the minimum fill level, and the calibrated vacuum ensures the tube fills to the correct draw volume when technique is correct. The PET plastic construction eliminates the glass breakage risk that was a significant hazard in coagulation tube handling. The Hemogard closure prevents finger contact with blood on the stopper surface during opening — a relevant safety feature in a haematology or coagulation laboratory where multiple tubes are opened per batch. For laboratory supply distributors servicing hospitals with active coagulation and haematology departments — the BD blue citrate tube is the first specification listed on any coagulation reagent kit's collection requirements. The 2.7ml format handles the majority of adult draw situations. Sara Wellness exports the full BD Vacutainer range. Standard 100/box and 1,000/case quantities available on both 1.8ml and 2.7ml variants.
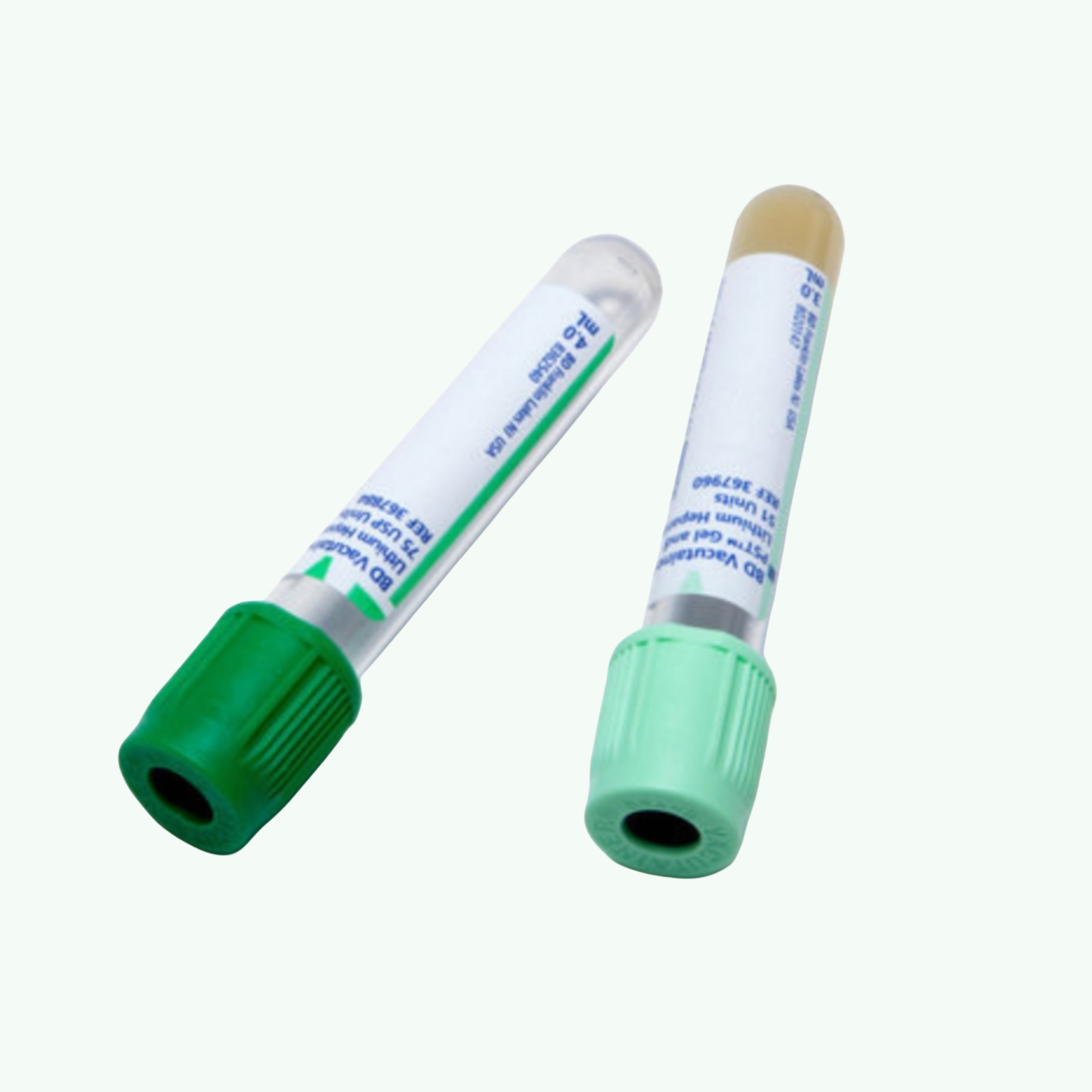
BD Vacutainer Heparin Tube 4ml (Green — Lithium Heparin, Plasma Chemistry)
There is a category of laboratory test request where waiting 30 minutes for blood to clot — as you would with a serum tube — is simply not the right answer. STAT electrolytes on a critically unwell patient. Ammonia levels that degrade rapidly. Plasma specimens for tests where clot activator or gel might interfere with the specific assay chemistry. These situations exist, they are common enough in busy clinical chemistry departments, and the green heparin tube is the solution. The BD Vacutainer Heparin Tube contains lithium heparin as its anticoagulant — sprayed directly onto the tube walls in precise concentration. Heparin works by activating antithrombin III, which inactivates thrombin and other clotting factors, completely preventing clot formation. The result is a plasma sample available as soon as the tube is centrifuged, with no clotting time required. No 30-minute wait. The analyser gets its sample faster. Lithium heparin is the preferred formulation over sodium heparin for the simple reason that sodium (introduced with sodium heparin) would interfere with plasma sodium measurements — and sodium is on virtually every basic metabolic panel. Lithium, on the other hand, is not a routine clinical chemistry analyte in standard panels, making lithium heparin the chemically neutral choice. The exception is therapeutic lithium drug level monitoring: lithium heparin tubes are contraindicated for plasma lithium level testing. The 4ml volume in a 13x75mm format sits in the practical zone for most routine clinical chemistry panels — enough volume for the full basic metabolic panel, liver enzymes, calcium, and phosphate, with a small reserve. The green Hemogard closure gives a clear visual identification that distinguishes plasma heparin tubes from the gold SST serum tubes they often run alongside in a chemistry batch. The PST variant — which adds a polymer gel separator to the lithium heparin formulation — produces a plasma tube with the same gel-barrier convenience as the gold SST for serum. After centrifugation, the gel migrates to separate plasma from cells, allowing direct aspiration without aliquoting. For laboratory supply distributors and hospital procurement managers — lithium heparin tubes are a steady, high-frequency consumable across every clinical chemistry department. Sara Wellness exports BD Vacutainer heparin tubes in both standard and PST gel formats, in box and case quantities, to wholesale buyers internationally.

BD Vacutainer EDTA Blood Collection Tube (Lavender — K2EDTA, Haematology)
Ask any haematology laboratory technician what is the single tube they cannot run out of, and the answer is always the same. Lavender top. K2EDTA. The CBC tube. Because the complete blood count is not simply the most ordered single blood test — it is the test that catches conditions nobody else catches. It is the peripheral blood film that shows the blast cells in acute leukaemia before any other test is abnormal. It is the haematocrit that diagnoses the quietly developing anaemia in a patient who came in complaining of fatigue. The lavender tube is where that diagnostic work begins. BD Vacutainer EDTA tubes contain K2EDTA — dipotassium ethylenediaminetetraacetic acid — as a dry spray-coat on the interior tube walls. EDTA chelates calcium ions, removing the calcium that the coagulation cascade absolutely requires at multiple steps. No calcium, no clot. The blood stays liquid. The red cells stay intact as individual biconcave discs. The white cells retain their nuclear and cytoplasmic morphology. The platelets remain as individual countable particles rather than forming aggregates. This is why EDTA is the anticoagulant of choice for haematology and not heparin. Heparin causes leukocytes to clump — making differential counting unreliable and blood film morphology assessment misleading. EDTA does not have this effect. At 1.8 mg/ml (the ISO 6710 standard concentration), the dry spray-coat formulation does not dilute the sample the way a liquid K3EDTA formulation would, which means MCV, MCHC, and haematocrit values are not affected by sample dilution. Apart from CBC, the lavender tube handles HbA1c (the glycated haemoglobin test that monitors three months of blood glucose control in diabetic patients), reticulocyte counts, haemoglobinopathy screening for sickle cell and thalassaemia, and blood grouping and crossmatch. For molecular testing, EDTA is the standard anticoagulant for blood DNA extraction because it inhibits nucleases that would degrade nucleic acids. For laboratory supply distributors covering hospital blood banks, haematology units, and clinical pathology laboratories — the EDTA lavender tube is the highest-volume item on the tube order list. Every active inpatient ward and every outpatient phlebotomy station consumes them continuously. Sara Wellness exports BD Vacutainer EDTA tubes in the full volume range to wholesale buyers. Box quantities and full case quantities both available.

Bilirubin Reagent Kit (Total and Direct — DPD Method, Clinical Biochemistry)
Bilirubin has a story. It starts in the spleen and bone marrow, where old red blood cells are broken down and their haemoglobin is metabolised into unconjugated bilirubin. That bilirubin travels to the liver, where it is conjugated with glucuronic acid to become water-soluble (direct bilirubin) and excreted into bile. When this process goes wrong — when the liver is failing, when bile ducts are blocked, when red cells are being destroyed faster than the liver can process them — bilirubin accumulates. The skin and eyes turn yellow. The bilirubin reagent kit is the laboratory's tool for quantifying how wrong things have gone. Bilirubin Reagent Kits use the DPD (3,5-dichlorophenyl diazonium tetrafluoroborate) method for total bilirubin measurement. The DPD method works by having the diazonium salt in Reagent 2 react with bilirubin in an acidic buffer environment to form azobilirubin — a coloured compound whose absorbance at 540nm is directly proportional to the bilirubin concentration in the sample. Direct bilirubin (conjugated) reacts without an accelerator; unconjugated bilirubin requires an accelerator (surfactant/solubiliser). Total bilirubin is measured by measuring both fractions together with an accelerating surfactant in Reagent 1. The DPD method has advantages over the classic Jendrassik-Grof method for automated analyser use: the R1/R2 two-reagent format is directly loadable on standard clinical chemistry analysers without modification, the reagents are stable in liquid ready-to-use form, and the method shows good correlation with reference methods validated against NIST SRM 916a bilirubin standard. Practical applications are broad: liver function testing in adults; neonatal jaundice monitoring (the highest-volume bilirubin testing setting globally — every newborn in most countries has bilirubin measured within 24-72 hours of birth); monitoring haemolytic anaemia; post-operative liver assessment; diagnosis of bile duct obstruction and cholestasis. For laboratory reagent distributors, hospital pharmacy buyers, and diagnostic supply importers — bilirubin reagent kits are a steady-volume biochemistry consumable across every clinical laboratory with a chemistry analyser. Sara Wellness exports clinical biochemistry reagents to international wholesale buyers with flexible order quantities.

Borosil Conical Glass Beaker Graduated (Borosilicate 3.3 — Laboratory Use)
The glass beaker is arguably the most recognisable piece of laboratory equipment in existence — more so than even the Erlenmeyer flask or the test tube. It appears on the covers of science textbooks, in the logos of chemistry departments, in every school laboratory from kindergarten to postgraduate level. This recognition is not incidental. The beaker has remained essentially unchanged in design for over a century because its design is, within its intended function, optimal. Borosil Conical Glass Beakers are manufactured from borosilicate 3.3 glass — the material designation that distinguishes laboratory-grade from ordinary glass. Borosilicate 3.3 has a coefficient of thermal expansion of 3.3 x 10-6 K-1, which is approximately one-third that of standard soda-lime glass. This low expansion means the glass resists thermal shock: a borosilicate beaker can go from a cold bench to a hot plate, or be heated rapidly with a Bunsen flame, without cracking from the differential expansion stresses that would shatter ordinary glass. In continuous service, borosilicate beakers handle heating to 230°C repeatedly without degradation, and can withstand a single extreme heat event to 490°C. The Borosil brand is manufactured by Borosil Scientific, an Indian laboratory glassware manufacturer with decades of production experience supplying borosilicate labware to Indian and international laboratory markets. Borosil beakers comply with IS 2619 (Indian Standard), ISO 3819 (international standard for laboratory glassware — beakers), and DIN 12331 (German standard) — the three major standards that define beaker dimensions, graduation marking, and material requirements for professional laboratory use. The white printed graduations are for reference volume estimation during routine use — not for accurate volumetric measurement, for which calibrated volumetric flasks and pipettes are the correct choice. The pouring spout at the beak allows controlled transfer of liquids without drips. For laboratory supply distributors, educational institution buyers, hospital laboratory supply procurement, and industrial laboratory buyers — Borosil graduated beakers are a genuine Indian-manufactured alternative to imported European laboratory glassware, certified to the same international standards, at competitive export pricing. Sara Wellness exports Borosil laboratory glassware to international wholesale buyers in multiple sizes and pack quantities.

Borosil Flat Bottom Amber Culture Tube
There is a particular kind of frustration that comes from losing a light-sensitive sample to an ordinary clear glass tube. The specimen is perfectly collected, the reagent is perfectly prepared, and then the tube fails the most basic job it was hired to do. Amber glass culture tubes exist precisely to prevent that particular catastrophe. Borosil Flat Bottom Amber Culture Tubes are made from borosilicate 3.3 glass, which is the same low-expansion composition used in scientific glassware worldwide for applications that demand chemical resistance and thermal stability. The amber colour is not a coating. It is fused into the glass, which means it does not peel, scratch off, or fade with repeated use or autoclaving. This matters enormously when you are storing photosensitive cultures, enzyme preparations, bilirubin solutions, or any specimen where UV and visible light exposure will degrade the contents over time. The flat bottom is important too. Flat-base tubes stand upright independently without a rack, which simplifies bench work during routine mixing, decanting, or observation. The tube mouth is fire-polished and smooth, accepting standard rubber stoppers and serum caps for contamination-free storage between processing steps. These tubes are designed for culture work, routine laboratory sample storage, teaching and training labs, and any application where the sample must be protected from light degradation. They autoclave reliably, rinse clean, and do not introduce contaminants into sensitive preparations. For laboratory supply distributors and procurement officers sourcing glassware at scale, Borosil is the benchmark Indian manufacturer whose products meet IS 4163 and ISO 4142 standards. The quality is consistent lot to lot, which eliminates the variation problems that plague lower-cost alternatives. Sara Wellness has spent 15 years building export partnerships with laboratory distributors across Asia, Africa, the Middle East, and beyond. Our pricing holds up at volume. MOQs are flexible enough to accommodate both trial orders and full-container shipments. Every consignment is documented correctly for smooth customs clearance, because a shipment that sits in port is not a shipment that earns repeat orders.

Borosil Round Bottom Amber Culture Tube
Not everything in a laboratory needs to stand on its own. Sometimes the round bottom is actually the right shape for the job, particularly when you need efficient heat transfer from a water bath, smooth sedimentation of particulates, or vortex mixing without dead zones at the corners that flat-based tubes inevitably create. Borosil Round Bottom Amber Culture Tubes deliver light protection and thermal performance in the curved-base format that certain laboratory workflows specifically require. The amber borosilicate 3.3 glass blocks UV and damaging visible wavelengths, protecting contents that degrade under ordinary laboratory light. Centrifugation, water bath incubation, and vortex mixing all work more efficiently in round-bottom geometry. The glass is borosilicate 3.3 throughout. That means chemical resistance to most acids, bases, and organic solvents. It means autoclave compatibility for repeated sterilisation cycles. It means dimensional consistency batch to batch, which matters when you are running assays where tube geometry affects sedimentation rates or mixing dynamics. These tubes are widely used in microbiology culture work, serology, haematology, pharmaceutical stability studies, and any setting where photosensitive samples need to sit in incubators or water baths for extended periods. The amber colour prevents light-catalysed reactions from distorting your results while the round bottom ensures even heat distribution. Borosil manufactures these to IS 4163 and ISO 4142 standards. The manufacturer has been India's leading scientific glassware producer for decades, and the consistency they deliver is why laboratory procurement departments across the world specify Borosil by name rather than by specification. Sara Wellness exports Borosil laboratory glassware to distributors and institutional buyers worldwide. We work directly with importers, not through layers of middlemen, so our pricing stays competitive and communication stays clear. Tell us the size mix you need and the quantity per shipment, and we will build a supply programme that makes reordering straightforward.

Borosil Culture Tube Flat Bottom with Screw Cap
A rubber stopper does its job. But there are situations where a screw cap does it better. Shipping samples across cities. Storing specimens overnight. Running long-term cultures without the rubber drying out. Any scenario where you need the closure to stay closed without constant supervision. Borosil Flat Bottom Culture Tubes with Screw Cap combine the dimensional precision and chemical resistance of borosilicate 3.3 glass with a leak-proof threaded polypropylene cap. The result is a tube you can store on its side in a refrigerator, hand to a courier, or leave in an incubator for 72 hours without worrying about evaporation or contamination. The flat bottom gives it stability. This tube stands on the bench without a rack, which is a minor convenience that becomes a significant time-saver across a hundred daily handling events. The glass is Borosil borosilicate 3.3, so it tolerates autoclaving, chemical cleaning, and repeated sterilisation cycles without degrading. The screw cap is polypropylene, which is separately autoclavable and chemically inert. These tubes are used heavily in microbiology, clinical sample collection, pharmaceutical preparation, and any laboratory context where sample integrity during transport or prolonged storage is non-negotiable. The threaded closure creates a positive seal without the user needing to push, twist, and wonder whether the stopper actually sealed. Borosil produces these to IS 4163 standards with consistent wall thickness and mouth dimensions. When you buy 10,000 tubes from this manufacturer, tube 9,997 fits the cap the same way tube 3 did. Sara Wellness exports Borosil laboratory glassware to wholesalers, medical distributors, and institutional procurement departments on every populated continent. We have 15 years of export documentation experience so your shipment clears customs on time. We price competitively for volume orders and we communicate in plain language, not in sales brochure language.

Borosil Culture Tube Round Bottom with Screw Cap
If the round bottom culture tube is the workhorse of centrifuge-driven laboratory workflows, then the version with a screw cap is the one you take on the road. Culture tubes get moved. They go into courier bags, shipping containers, field collection kits, and hospital transport systems. A stopper that can work loose under pressure is not an acceptable closure for any of those environments. Borosil Round Bottom Culture Tubes with Screw Cap pair the curved-base geometry that centrifuge and water-bath work demands with a threaded polypropylene closure that locks positively and stays locked. No stopper anxiety. No evaporation surprises. No contamination from poorly-seated rubber during overnight incubation. The glass is borosilicate 3.3, manufactured by Borosil Scientific, the original Indian borosilicate glassware manufacturer. Chemical resistance is broad. Thermal resistance covers autoclave sterilisation cycles repeatedly. The round base gives smooth vortex mixing and even sedimentation. The screw cap gives you a seal you can trust without checking it every hour. Applications span a wide range: microbiology transport tubes, serology specimen collection, pharmaceutical stability studies, fermentation research, and clinical laboratory specimen processing where samples move between departments or facilities. The round bottom geometry is also useful in gravity filtration setups where the tube collects filtered liquid without a flat surface to catch unwanted particulate aggregation. Borosil manufactures these consistently, to the same dimensions, same glass composition, and same threading tolerances across every production lot. Sara Wellness has been supplying Borosil laboratory glassware to international buyers for 15 years. Our export process handles documentation, compliance certification, and consolidated shipments that let you order multiple Borosil lines in one container. We deal one-on-one with each client. Ask us about pricing, and you will get a number, not a brochure.

Borosil Glass Beaker Graduated (Low Form)
The beaker is arguably the most used piece of laboratory glassware in existence. It holds things. It mixes things. It heats things. It collects things. And when it fails, it usually does so in a dramatic and irreversible way at the worst possible moment. Which is why the glass it is made from is not a trivial detail. Borosil graduated glass beakers are made from borosilicate 3.3, the low-expansion glass composition that handles thermal shock without drama. You can take one from a cold bench and put it directly on a hot plate. You can rinse it with boiling buffer and then cold water in the same motion. The glass does not crack from the temperature change because the thermal expansion coefficient is low enough to absorb it. The low-form Griffin design is what you reach for by default. Wide diameter relative to height gives you a large, stable base that does not tip over when you bump the bench. The wide opening makes stirring, adding reagents, and pouring practical. Graduated markings are white printed and provide reference volume estimates — not calibrated measurements, but accurate enough for preparation and dilution work where a pipette will handle the precision later. Borosil manufactures these beakers to IS 2619 / ISO 3819 / DIN 12331 standards. The glass is chemically resistant to most laboratory acids, bases, and organic solvents, with the standard exceptions of hydrofluoric acid and hot concentrated alkalis. Every beaker is autoclavable. For laboratory equipment distributors sourcing Indian-manufactured scientific glassware, Borosil is the name that removes the quality question entirely. The brand is known globally and is specified by name in procurement lists from university labs to pharmaceutical production facilities. Sara Wellness has 15 years of experience exporting Borosil glassware in consolidated shipments to buyers who mix multiple product lines in one order. We keep the paperwork straight, the pricing honest, and the communication simple. Contact us for a bulk quotation.

Borosil Glass Reagent Bottle
Reagents are expensive. The time spent preparing them is expensive. The experiments they enable are expensive. So storing them in a bottle that leaks, reacts with the contents, or admits light when it should not is a particularly avoidable problem. Particularly when the alternative costs only slightly more. Borosil Glass Reagent Bottles are made from borosilicate 3.3 and closed with ground glass stoppers or polypropylene screw caps depending on the variant. The glass is chemically inert to an exceptional range of laboratory chemicals — acids, bases, organic solvents, buffer solutions, stains, and indicator solutions. It does not leach plasticisers, does not absorb organic compounds onto its surface, and does not react with anything except hydrofluoric acid and strongly concentrated hot alkalis. The wide-mouth design makes filling, pouring, and cleaning practical. Narrow-mouth variants are available for volatile solvents where minimising vapour loss during dispensing matters. Ground glass stoppers create an airtight seal for reagents that absorb moisture from air or release vapours at room temperature. The polypropylene screw-cap variants are autoclavable and suited to aqueous solutions, buffers, and culture media. These bottles are used in every serious laboratory context: pharmaceutical QC, university chemistry, clinical biochemistry, food testing, environmental analysis, and industrial quality control. The 60ml size suits small-batch reagent preparation. The 5000ml size suits bulk preparation of buffer systems or staining solutions used in high-volume processing. Borosil manufactures reagent bottles to IS 1388 and ISO 4800 standards. Consistency is the reason the brand has been specified by name in institutional purchasing contracts since before most current lab managers started their careers. Sara Wellness exports Borosil reagent bottles globally. We handle mixed-SKU container orders, export documentation, and quality certification. Flexible MOQs make it practical to trial new sizes without over-committing inventory.

Borosil 1000ml Reagent Bottle Screw Cap
The litre-sized reagent bottle is the workhorse container of any production-volume laboratory. Buffer solutions, staining reagents, wash solutions, culture media — everything that gets prepared in bulk and dispensed repeatedly across a working day starts here. The question is not whether you need a 1000ml reagent bottle. It is whether the one you are buying will still be reliable in cycle 200 of autoclaving. Borosil's 1000ml Reagent Bottle with polypropylene screw cap is the answer for labs that run through reagents at volume and cannot afford containers that cloud, crack, or fail. The glass is borosilicate 3.3. The screw cap is polypropylene. Both are autoclavable. The combination delivers a container that sterilises cleanly, seals tightly between uses, and resists virtually every reagent you will put inside it. The 1000ml size is available in both wide-mouth and narrow-mouth formats. Wide mouth for reagents that you pour, scoop, or dispense generously. Narrow mouth for organic solvents where you want to minimise vapour loss during the pour. The screw cap mechanism is straightforward and positive, without the alignment dependency of ground glass stoppers that can sieze or chip if not handled carefully. This is specifically the bottle that microbiology labs reach for when preparing a litre of LB broth. That biochemistry labs fill with phosphate buffer every Monday. That hospital pathology departments use for haematoxylin staining solution. High-volume, repeat-fill, repeat-autoclave applications where the container needs to perform identically on the hundredth use as it did on the first. Borosil manufactures this product to IS 1388 and ISO 4800. The Borosil name carries immediate recognition in global laboratory procurement. Sara Wellness ships Borosil 1000ml reagent bottles to laboratory distributors and institutional buyers worldwide. Volume pricing available. Export documentation handled end-to-end.

Borosil Glass Stirring Rod 205mm
It is a rod. It stirs. That sentence covers perhaps 10% of what a laboratory stirring rod actually does. A glass stirring rod scrapes precipitates off beaker walls. It transfers the last drop of a precious reagent. It aids filtration by directing liquid flow onto filter paper without splashing. It breaks up clumps in suspensions. It checks viscosity by how the liquid moves when pulled. And it does all of this inside chemical environments that would dissolve a metal rod, contaminate a reaction, or melt a plastic one. Borosil's 205mm glass stirring rods are manufactured from borosilicate 3.3 rod stock and fire-polished at both ends to remove sharp edges. The 205mm length fits comfortably across a 250ml to 1000ml beaker without being unwieldy. Common diameters are 5mm, 6mm, and 8mm — the wider the rod, the more mixing force per stroke and the more physically robust the rod is for heavy-duty stirring applications. Borosilicate glass is the only sensible material for a general-purpose laboratory stirring rod. It is chemically inert to almost everything a routine analytical or preparative lab will encounter. It does not react with acids, alkalis, organic solvents, or buffer solutions. It can be heated if necessary. It autoclaves without distortion. It can be cleaned with any standard laboratory glassware detergent and returned to service in the same condition it started in. These are used in chemistry, biochemistry, biology, pharmaceutical, clinical, and food testing laboratories everywhere on earth. They are so fundamental to laboratory work that procurement departments sometimes treat them as consumable stationery. Which is all the more reason to buy them from a manufacturer whose dimensional consistency means the rod fits the beaker, the rubber policeman fits the rod, and everything works as it should. Sara Wellness exports Borosil laboratory accessories to institutional buyers and laboratory distributors globally. Simple pricing. Reliable supply. Fast response.

Borosil Tall Glass Beaker Graduated (Berzelius)
Most laboratories own two types of beakers and use the same one for everything. The short one sits on the bench, does the mixing, holds the liquid, and collects the splash. The tall one stands in a corner waiting for the rare procedure where a short beaker would actually fail. That rare procedure happens more often than you think. Gravity filtration is one. When you want liquid to drip through a funnel into a container below, a tall beaker collects the filtrate without splashing it back onto the funnel or getting in the way of the setup. Precipitation reactions are another — a taller column of liquid keeps the precipitate from re-mixing with the supernatant when you are trying to observe sedimentation clearly. Borosil Tall Form (Berzelius) Graduated Beakers are manufactured from borosilicate 3.3 glass. The taller, narrower profile compared to the standard low-form Griffin beaker creates a longer liquid column, easier observation of settled material or layered solutions, and a more stable fit under filtration funnels. Graduated markings are reference scale with a typical ±5% tolerance. Pouringspout and polished rim are standard features. Chemical resistance covers the same ground as all borosilicate 3.3 glassware: broad resistance to acids, organic solvents, aqueous solutions, and buffer preparations. Autoclavable. Cleanable with standard laboratory glassware detergents. Compatible with magnetic stir bars if a compatible size is used. For lab distributors selling to filtration-heavy applications, titrimetric analysis, and analytical chemistry departments, this product rounds out any complete Borosil beaker range without needing to source from a different manufacturer. Sara Wellness handles Borosil export orders mixing low-form, tall-form, and speciality beakers in the same consignment. Fifteen years of export logistics means your mixed order arrives documented, certified, and on time.

Bovine Albumin 10ml (BSA Serology Reagent)
Blood grouping is a procedure where being almost right is precisely as useful as being entirely wrong. An incorrect crossmatch sends a patient the wrong blood. The consequences of that error are severe, fast, and irreversible. Which is why every reagent in the blood bank — including the ones that seem basic — needs to be exactly what it claims to be. Bovine Albumin in the 22% and 30% formulations is an enhancement medium used in blood banking and serology. It potentiates the agglutination of red blood cells sensitised with incomplete antibodies (primarily IgG class), allowing reactions to be detected by indirect antiglobulin techniques and tube methods that would otherwise produce weak or invisible results. Without an enhancement medium, many clinically significant antibodies simply do not produce readable agglutination under routine conditions. This 10ml vial format is the standard size for blood bank use. The concentrated albumin solution is added to the test system at the appropriate volume per the procedure in use. Both 22% and 30% concentrations are available because different blood banking protocols specify different albumin concentrations for optimal enhancement effect. The reagent is manufactured under strict quality controls, with defined protein concentration, appropriate pH, and preservatives that maintain stability throughout the stated shelf life. Lot-to-lot consistency is essential in blood banking — a reagent that performs differently between lots introduces an unacceptable variable into a high-stakes clinical procedure. For blood bank reagent distributors and hospital procurement teams sourcing serology reagents at volume, this is a well-understood, widely used product with predictable consumption rates tied directly to blood bank test volume. Sara Wellness exports blood banking and serology reagents from India, working with distributors and hospital groups across export markets. We handle the cold chain documentation, import certification requirements, and quality records that blood bank reagent procurement demands.

Amylase Reagent 60ml Liquid Stable (CNPG3 Method)
Acute pancreatitis has a signature. It presents with sudden severe abdominal pain, and the blood test that confirms the clinical suspicion is a serum amylase measurement. Get that result fast and accurately, and the clinical team can act. Get it slowly or inaccurately, and the patient waits while the pancreas continues to be a problem. Amylase reagent is the biochemical consumable that makes that measurement possible. The liquid-stable, 60ml format of CNPG3 method amylase reagent is designed specifically for clinical biochemistry laboratories that need a ready-to-use, on-board-stable reagent compatible with automated chemistry analysers — the kind of setup where 50 amylase tests run before 9 AM and every result needs to be right. The CNPG3 substrate (2-chloro-4-nitrophenyl-alpha-D-maltotrioside) is the modern enzymatic substrate for amylase quantification. The reaction is direct, sensitive, and highly specific for amylase activity in serum and urine. No turbidimetry variables, no long incubation periods, no reagent reconstitution. The liquid-ready format goes directly onto the analyser. On-board stability allows the reagent to sit loaded in the instrument for weeks without degrading, which is critical for labs that run amylase frequently but not on every batch. The 60ml volume calibrates conveniently for analysers running 100-200 tests per bottle. Results are traceable to IFCC reference method, ensuring inter-laboratory comparability. For laboratory reagent distributors and hospital procurement departments building clinical chemistry reagent programmes, amylase is a core test that rarely gets removed from the panel — which means predictable, recurring demand. Sara Wellness exports clinical chemistry reagents to laboratory distributors and hospital procurement teams worldwide. We know the cold chain documentation these shipments require, we manage the logistics accordingly, and our pricing is built for B2B volume, not retail margins.
Cholesterol LDL Direct Liquid Stable Reagent
LDL cholesterol occupies a specific clinical position. It is the lipoprotein fraction most directly associated with atherosclerotic cardiovascular risk, which makes it the number that cardiologists, internists, and primary care physicians watch most closely when managing a patient's lipid profile. It is also the number that used to require a fasting specimen and a calculation involving three other results. The Friedewald equation worked until it did not, and most laboratories have moved on. The direct LDL cholesterol assay removes the fasting requirement and eliminates the calculation entirely. It measures LDL-C directly in the serum or plasma sample using a two-reagent enzymatic system. Detergents selectively solubilise non-LDL lipoproteins (HDL, VLDL, chylomicrons) in R1, leaving LDL intact. R2 then solubilises the LDL fraction and the cholesterol is measured enzymatically at 600-700nm. The direct method is not thrown off by hypertriglyceridaemia, which was the Achilles heel of the Friedewald approach. The liquid-stable format is what makes it practical for routine laboratory operation. Both R1 and R2 are ready-to-use solutions requiring no reconstitution. On-board stability at analyser operating temperatures (37°C) allows the reagent to remain loaded for extended periods without degrading, which matters in a high-throughput biochemistry lab where the lipid profile panel runs continuously. Compatibility spans semi-automated and fully automated clinical chemistry analyser platforms, with application parameters available or programmable for most common systems. Sara Wellness exports clinical chemistry reagents including LDL direct, total cholesterol, and HDL cholesterol reagents to laboratory distributors and hospital procurement teams internationally. Cold chain logistics are part of our standard export process for these products. Pricing is built for volume B2B relationships, not individual test pricing.

Cholesterol Reagent Kit (Total Cholesterol CHOD-PAP)
Every laboratory that runs a lipid panel runs total cholesterol. It is the first number in the panel, the anchor for calculating derived values, and one of the most frequently requested tests in clinical biochemistry. Which means the reagent you use for it runs through your analyser in volume. Consistency matters more here than in tests run occasionally. The CHOD-PAP (Cholesterol Oxidase-Peroxidase, Phenol-4-Aminoantipyrine) enzymatic colorimetric method has been the clinical chemistry standard for total cholesterol measurement for decades. The chemistry is well-characterised, calibrated against accepted reference standards, and produces results that compare reliably across laboratories and analyser platforms. The reaction reads at 505nm, which falls cleanly within the photometric range of every clinical chemistry analyser in use. This reagent kit is a single-reagent, liquid-stable system. That means: pour it on the analyser, load the calibrator, run the controls, process samples. No reconstitution steps, no waiting for lyophilised powder to dissolve and equilibrate. Linear range extends up to 750 mg/dL under standard conditions, covering the full clinically relevant spectrum including severely hypercholesterolaemic patients. Pack sizes range from 25ml two-vial formats suitable for low-volume or semi-automated work, through to 500ml and 1000ml formats for high-throughput automated systems where reagent loading frequency is a real operational consideration. For laboratory reagent distributors, total cholesterol is a guaranteed reorder item tied to laboratory test volume. A laboratory that runs 200 lipid panels per day is going to order cholesterol reagent on a very predictable schedule. That predictability is good for supply chain planning on both sides. Sara Wellness exports clinical chemistry reagents with appropriate cold chain documentation to laboratory distributors across export markets.
CK-MB Liquid Stable Reagent (Immunoinhibition Method)
When a patient presents with chest pain, the clock starts immediately. The clinical team needs troponin, ECG, and CK-MB — fast. The value of CK-MB in acute myocardial infarction workup is its time course: it rises within 4-6 hours of an infarct, peaks at 12-24 hours, and returns to normal within 3-4 days. That early rise and relatively clear temporal pattern is what makes it useful for both diagnosis and timing of the infarct event. CK-MB liquid stable reagent uses the immunoinhibition method. M-subunit activity (CK-MM and the M component of CK-MB) is inhibited by anti-M antibodies in R1. The remaining activity measured after inhibition represents the B-subunit contribution, which in myocardial tissue means CK-MB activity. The result is expressed as CK-MB activity in IU/L. The liquid-stable, two-reagent format is the practical choice for any laboratory running cardiac markers at volume. Both R1 and R2 arrive ready to use, load directly onto the analyser, and remain stable on-board without daily preparation steps. The chemistry is proven and widely accepted across clinical biochemistry guidelines for acute coronary syndrome workup. For emergency departments and cardiac care units that run CK-MB around the clock, reagent consistency is not a preference. A batch of reagent that performs differently from the last affects clinical decisions on patients who cannot wait for an investigation into why the numbers shifted. Sara Wellness exports cardiac marker reagents including CK-MB to laboratory distributors and hospital procurement departments internationally. We understand the cold chain and documentation requirements for these products and manage them as part of every export shipment.
CK-NAC Liquid Stable Reagent (Creatine Kinase UV Kinetic)
Creatine kinase sits at an interesting crossroads in clinical biochemistry. Total CK is the initial alarm. It goes up whenever muscle is damaged, which covers a spectrum from athletic overexertion to myocardial infarction to rhabdomyolysis to the side effects of certain medications. That breadth of elevation is useful for screening but demands further investigation. CK-NAC is the accurate, standardised measurement of total CK activity that makes that screening trustworthy. CK-NAC uses the UV kinetic method recommended by both the International Federation of Clinical Chemistry (IFCC) and the German Society for Clinical Chemistry (DGKC). N-acetyl-L-cysteine (NAC) activates the CK enzyme and increases reaction efficiency. The reverse reaction format (measuring the conversion of creatine phosphate and ADP to creatine and ATP, coupled to NADPH production) produces a rate of absorbance change at 340nm that is directly proportional to CK activity. The reagent is liquid-stable, supplied as a two-component system (R1 and R2) that mixes in a defined ratio to produce the working reagent. On-board stability allows the prepared working solution to remain usable for up to 10 days at 2-8°C, reducing the preparation frequency on busy automated platforms. Linearity extends to 2000 IU/L, which covers even significantly elevated CK values without requiring dilution in most patient scenarios. For clinical laboratories serving emergency departments, cardiac care, sports medicine, and neurology wards, CK-NAC runs continuously. It is not an esoteric panel — it is a daily volume test in any biochemistry laboratory with significant acute care throughput. Sara Wellness exports clinical chemistry reagents to diagnostic laboratory distributors worldwide.

Clot Activator Vacuum Blood Collection Tube
The pre-analytical phase of laboratory testing is responsible for the majority of laboratory errors. Not the analyser, not the reagent, not the calibration. The sample itself — how it was collected, what tube it was collected in, whether it clotted correctly and fully before centrifugation. A serum tube that does not clot properly yields an unreliable specimen, and an unreliable specimen produces a result that looks real but is not. Clot activator vacuum blood collection tubes solve the clotting part of that problem. The inner wall of each tube is coated with microscopic silica particles (or a spray-dried thromboplastin in some variants) that activate the coagulation cascade on contact with blood, accelerating clot formation to approximately 30 minutes compared to the 60 minutes or more that plain (non-additive) glass tubes require. Consistent, complete clot formation before centrifugation is what produces clean serum that the analyser reads accurately. The plastic PET tube construction makes them virtually unbreakable compared to glass, which matters in sample transport and in phlebotomy settings where dropped tubes are an occupational reality. The tubes are evacuated to draw the labelled volume, eliminating the need for the phlebotomist to estimate fill volume. Red cap or gold cap variants are available, with or without a gel separator that forms a stable barrier between serum and cells after centrifugation. For laboratory consumable distributors, vacuum blood collection tubes are a daily-use consumable with consumption directly proportional to patient sample volume. Every serum biochemistry test requires one. Every lipid panel, liver function, kidney function, or endocrine workup starts with this tube. Sara Wellness exports blood collection tubes to laboratory distributors and hospital supply chains internationally.

Creatinine Reagent Kit (Jaffe Kinetic Method)
Creatinine is the most frequently ordered renal function test in clinical biochemistry. Every renal panel, every metabolic panel, every critical care patient assessment includes it. In a busy laboratory running 200 renal panels a day, the creatinine reagent goes through the analyser continuously. Its behaviour — lot-to-lot consistency, on-board stability, calibration linearity — is not a feature; it is a prerequisite. The Jaffe kinetic method is the most widely used clinical chemistry approach for serum creatinine measurement. Creatinine reacts with alkaline picrate to produce an orange-red complex, and the kinetic measurement tracks the rate of colour change over a defined time window. The kinetic format (reading the rate of reaction rather than the endpoint colour) reduces interference from non-creatinine chromogens — the long-standing limitation of the Jaffe endpoint method. This creatinine reagent kit uses the modified kinetic Jaffe method calibrated to IDMS-traceable standards, which is the standardisation approach adopted following NKDEP and IFCC recommendations for inter-laboratory comparability. The modification aligns results more closely with the reference method values that are used to calculate eGFR (estimated glomerular filtration rate) using the CKD-EPI or MDRD equations. The reagent is liquid stable, supplied in a format ready for use on semi-automated and fully automated chemistry analysers. On-board stability at 37°C allows the reagent to remain loaded for the stated stability period without degradation. For laboratory reagent distributors, creatinine is a guaranteed volume item tied directly to sample throughput. It is also a quality-sensitive item — laboratory accreditation bodies check creatinine performance as part of external quality assessment. Sara Wellness exports clinical chemistry reagents with full cold chain documentation to laboratory distributors internationally.

CRP Test Kit 25 Tests (Microsidd)
There is a category of laboratory test that earns its place in every diagnostic setting from the most sophisticated reference laboratory to the most resource-limited rural clinic. The CRP latex test is one of them. It requires no instrument, no electricity, no cold chain for the test itself, and produces a result in three minutes that is as clinically useful in triage as a result that takes three hours on an automated analyser. C-Reactive Protein (CRP) is synthesised by the liver and released into the bloodstream in response to inflammation, infection, or tissue injury. Its serum concentration rises within hours of an acute inflammatory event and falls rapidly when the trigger resolves. This rapid kinetic makes it a useful marker for detecting active inflammation, monitoring treatment response, and differentiating bacterial from viral infection in clinical decision-making. Microsidd's CRP Test Kit uses the latex agglutination slide test principle — one of the most established and validated methods in clinical serology. Latex particles coated with anti-CRP antibodies agglutinate (clump visibly) when mixed with patient serum containing CRP above the detection threshold (typically 6 mg/L). The test is read visually under a light source after the three-minute reaction time. Positive agglutination is clear and unambiguous; negative results show no agglutination. Semi-quantitative titration is possible by diluting the sample and reading the endpoint. The 25-test kit format is the standard for smaller laboratories and clinic-based testing. Larger 50 and 100-test kits are available for higher-volume settings. For laboratory supply distributors, CRP latex kits are a steady consumption item across pathology labs, hospitals, and polyclinics in every export market. Sara Wellness exports diagnostic test kits including serology reagents to laboratory distributors internationally.

Dengue Day 1 Test Kit (NS1 Ag + IgM + IgG Combo Rapid Test)
Dengue is a disease that has a very particular problem: it looks like a lot of other things at the beginning. A patient presents with fever on day one or day two. Headache, myalgia, and the clinical concern about where this is going. The classic dengue rash may not have appeared. Laboratory confirmation is what separates dengue from the differential diagnosis list, and doing that confirmation from day one — before the antibody response has developed — is where the NS1 antigen test changes the picture. The Dengue Day 1 Test Kit by J. Mitra & Co. is a three-in-one rapid immunochromatographic test device that detects NS1 antigen (present from day one of infection), IgM antibodies (which develop in primary infection from around day four), and IgG antibodies (present in secondary infection and past exposure) simultaneously from a single patient serum or plasma sample. Two devices per test kit cover both the antigen and the antibody component, with the IgM/IgG device having three lines — C (control), M (IgM), and G (IgG). The result interpretation maps directly onto dengue diagnosis and infection type classification. NS1 positive in the early febrile phase indicates active infection. IgM positive suggests primary dengue; IgG positive alongside IgM or IgG alone suggests secondary infection or recent past exposure. The manufacturer's claimed performance figures for the kit are: NS1 Ag sensitivity 96%, specificity 98%; IgM/IgG antibody sensitivity 95%, specificity 97%. The kit detects all four dengue serotypes (DEN-1, DEN-2, DEN-3, DEN-4). Shelf life is 30 months at 2-30°C, which allows storage at ambient temperature in most tropical markets — important for clinics and hospitals in endemic regions without reliable cold chain at every access point. For diagnostic laboratory distributors operating in dengue-endemic markets across Southeast Asia, South Asia, Africa, and Latin America, the Dengue Day 1 test kit is a high-turnover product that aligns with the seasonal dengue case load in endemic settings. Sara Wellness exports J. Mitra diagnostic test kits to laboratory distributors internationally, with cold chain documentation and proper IVD classification for destination market regulatory compliance.

Dengue IgG Microlisa ELISA Test Kit (J. Mitra)
Rapid tests confirm dengue. ELISA defines dengue serologically with a precision and throughput that no rapid test can match. In a reference laboratory processing samples from a hospital system or a public health surveillance programme, the ELISA format is not a substitute for the rapid test — they serve different purposes. The rapid test answers a patient's question quickly. The ELISA answers an epidemiologist's or a blood bank's question thoroughly. Dengue IgG Microlisa from J. Mitra & Co. is a solid-phase enzyme-linked immunosorbent assay designed for the in-vitro qualitative detection of dengue-specific IgG antibodies in human serum or plasma. The test principle is capture ELISA: anti-human IgG antibodies are coated on the microwell surface. Patient samples added to the wells allow dengue-specific IgG to bind to these coated antibodies. After washing, HRPO-conjugated dengue antigen (DEN-4) is added, which binds to the dengue-specific IgG already captured in the well. Substrate addition produces a colour signal proportional to the dengue IgG concentration, read at 450nm. The IgG Microlisa detects all four dengue serotypes (DEN-1 through DEN-4). Performance figures published by J. Mitra for this kit stand at 99.93% specificity and 96.00% sensitivity — figures that place it in the high-performance tier for commercial dengue ELISA serology. Shelf life is 18 months at 2-8°C. The 96-well break-away microplate format allows laboratories to run partial plates when sample volumes are below 96 without wasting the unused wells. For reference laboratories, blood banks performing donor screening for dengue in endemic areas, and public health surveillance programmes, this ELISA kit is an established tool from a well-known Indian IVD manufacturer. Sara Wellness exports J. Mitra diagnostic products internationally. Cold chain documentation and IVD export classification are handled correctly for every shipment.

Dengue NS1 Ag Microlisa ELISA Test Kit (J. Mitra)
When a patient presents in the early febrile phase — day one or day two of fever — the dengue virus is actively replicating and the NS1 protein is being secreted into the bloodstream in measurable concentrations. The antibody response has not yet developed. This is the window where the NS1 ELISA is irreplaceable. It can detect what nothing else can at that moment: the virus itself, through its antigen. Dengue NS1 Ag Microlisa from J. Mitra & Co. is a direct sandwich ELISA for the in-vitro qualitative detection of dengue NS1 antigen in human serum or plasma. The assay geometry is direct sandwich: anti-dengue NS1 antibodies are coated on the microwell surface. The dengue NS1 antigen from the patient sample binds to these antibodies. Then HRPO-conjugated monoclonal anti-dengue NS1 antibodies are added, forming a sandwich complex around the captured NS1. Substrate produces a blue colour whose intensity is proportional to NS1 concentration, stopped and read at 450nm. The performance figures J. Mitra publishes for this kit are exceptional: sensitivity 99.5%, specificity 100%. A sensitivity of 99.5% means that of all confirmed dengue NS1-positive samples tested, 99.5% were correctly identified as positive. A specificity of 100% means no false positives were generated in the validation set. These are clinically significant numbers for an early dengue diagnostic test. All four dengue serotypes (DEN-1, DEN-2, DEN-3, DEN-4) are detected. Shelf life is 24 months at 2-8°C, which is a generous storage window for laboratory procurement planning. For blood bank screening (dengue can be transmitted through transfusion in endemic areas), reference laboratory confirmation, and public health surveillance programmes operating at scale, the 96-well ELISA format and the high-sensitivity performance of this kit make it a strong choice. Sara Wellness exports J. Mitra ELISA test kits to laboratory distributors internationally, with correct IVD export classification and cold chain documentation.

DiaSys QDx Urine Test DS 10 Reagent Strips
Urinalysis is the oldest diagnostic test in medicine. Physicians have been examining urine for clinical information for thousands of years. What has changed is not the basic utility of the test but the speed and format with which results are obtained. The modern reagent strip delivers 10 parameters in approximately 60-120 seconds from a single dip. For a busy outpatient clinic, an emergency department, or a rural health post where laboratory infrastructure is limited, this speed and simplicity is not a convenience — it is the difference between results at the point of care and results that arrive after the patient has gone home. DiaSys QDx Urine Test DS 10 Reagent Strips are 10-parameter urinalysis strips manufactured by DiaSys Diagnostics India Private Limited, the Indian subsidiary of DiaSys Diagnostic Systems GmbH (Germany). DiaSys India's manufacturing facility in Mahape, Navi Mumbai is ISO 9001:2015 and ISO 13485:2012 certified, and the QDx product range carries CE marking for international markets. The DS 10 strip detects 10 key urine parameters — typically glucose, protein, bilirubin, urobilinogen, pH, blood, ketones, nitrites, leukocytes, and specific gravity. The sensitivity of each parameter pad is calibrated to be comparable to or better than established market references according to DiaSys specifications. The colour chart printed on each pack allows visual reading without an analyser, making the strip usable in any clinical setting. For automated reading, the QDx DS 10 strips are compatible with the DiaSys QDx UriLyzer 100 Pro urinalysis analyser, which provides quantitative results (mg/dL) alongside qualitative readings, operates at throughput of up to 120 tests per hour in fast mode, and eliminates observer-dependent colour interpretation variability. The canister format includes a double desiccant to maintain strip integrity in humid environments — particularly relevant for tropical climates where single-desiccant packaging can compromise strip performance well before the printed expiry date. For laboratory equipment distributors and diagnostics supply companies, urinalysis strips are a high-volume consumable purchased in consistent quantities by every hospital lab, clinic, and health centre. DiaSys QDx strips offer an Indian-manufactured CE-marked option at pricing that reflects local production costs. Sara Wellness exports DiaSys QDx diagnostic products to laboratory supply distributors internationally.
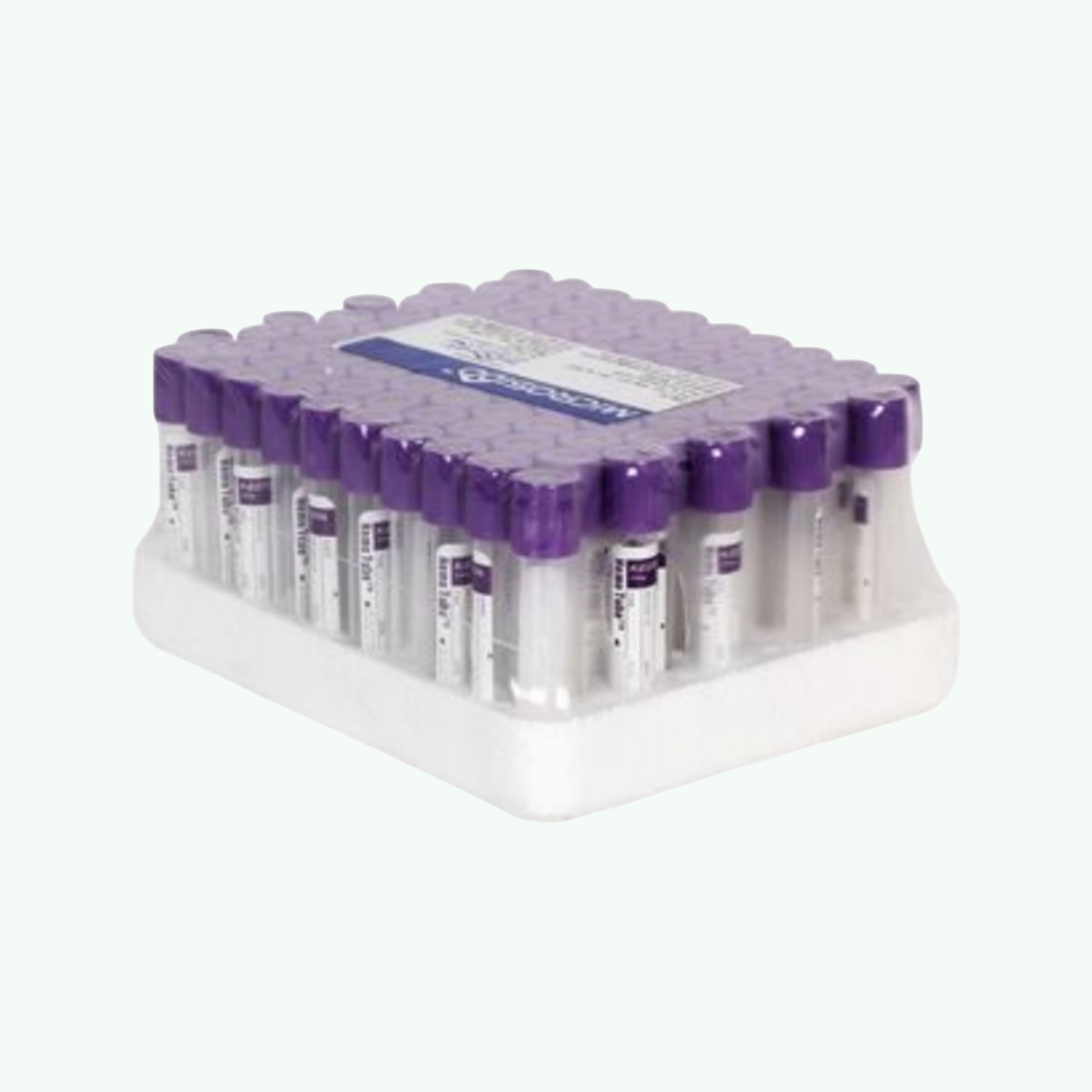
K3 EDTA Vacuum Blood Collection Tubes (Purple/Lavender Top)
Haematology investigations depend entirely on what happens to the blood sample between the vein and the analyser. Get the anticoagulant wrong, and the cells clump, the count is off, and the platelet figure is meaningless. The K3 EDTA vacuum collection tube exists precisely to prevent this — potassium tripotassium EDTA dissolves instantly into the drawn sample and chelates the calcium ions that the coagulation cascade requires, stopping clot formation before it can begin. K3 EDTA vacuum blood collection tubes (purple/lavender top) are the standard sample tube for complete blood count (CBC) investigations, haematological indices, blood film preparation, blood group and cross-match, and DNA-based molecular testing. The liquid K3 EDTA form provides faster dissolution in the sample than K2 EDTA (which is a dry spray coating) and is considered preferable for automated haematology analysers where any undissolved anticoagulant can cause interference. The tubes are manufactured as pre-evacuated glass or PET plastic cylinders with a predetermined negative pressure (vacuum) that draws a fixed, calibrated volume of blood when the needle enters the vein. The pre-measured EDTA content is matched to the tube's nominal draw volume so that the anticoagulant-to-blood ratio is consistent from tube to tube, which is essential for accurate haematological indices that are sensitive to dilution effects. The colour-coded lavender cap is the internationally recognised indicator for EDTA tubes across all major vacuum tube systems, making identification reliable in multi-specialty laboratory settings and emergency departments where multiple tube types are in use simultaneously. For laboratory supply distributors and hospital procurement offices sourcing phlebotomy consumables, vacuum blood collection tubes are one of the highest-volume purchase items in any clinical laboratory setting. Indian manufacturers supply EDTA tubes at significantly competitive pricing relative to imported European brands while meeting equivalent manufacturing standards. Sara Wellness exports laboratory consumables including vacuum blood collection tubes to medical distributors internationally.

K2 EDTA Vacuum Blood Collection Tubes (Purple/Lavender Top)
Collecting a blood sample correctly is the foundation of every laboratory result that follows from it. The tube, the anticoagulant, the draw volume, the mixing protocol — all of it matters. An error before the sample reaches the analyser is invisible. The result looks like a result. It is wrong, and no one in the laboratory chain of custody can know it without re-drawing. Pre-analytical quality is underappreciated precisely because its failures are silent. K2 EDTA vacuum blood collection tubes are the most widely used haematology collection tube in clinical laboratories globally. The K2 designation refers to dipotassium EDTA, supplied as a dry powder or spray coating on the inner wall of the tube. When blood enters the evacuated tube, the EDTA dissolves into the sample and immediately chelates the free calcium ions in the plasma, stopping the coagulation cascade at its earliest step and preserving the cellular elements in their original state for analysis. The internationally standardised lavender/purple cap identifies the tube as an EDTA tube across all phlebotomy systems, enabling accurate tube identification during multi-tube collection protocols where several different tube types are drawn from the same venepuncture. The draw order (sodium citrate before EDTA before serum tubes) is a specific requirement of phlebotomy protocols, and the colour-coding system supports this. K2 EDTA spray-coating provides a uniform anticoagulant distribution with no liquid volume added to the sample, which means the EDTA-to-blood ratio dilution effect is essentially zero compared to liquid K3 EDTA formulations. CLSI and ICSH guidelines cite this as the reason K2 is slightly preferred for automated haematology analysis where cell count reference ranges are calibrated against samples without dilution correction. For laboratory supply distributors, K2 EDTA tubes are one of the highest-frequency consumable items in any product catalogue. Sara Wellness exports laboratory phlebotomy consumables to medical distributors internationally.

Eryclone Anti-A,B Monoclonal Antibody Reagent (Blood Grouping)
Blood group determination is not optional when a transfusion is in play. Give a patient incompatible blood and you have initiated an acute haemolytic transfusion reaction — one of the most dangerous acute events in clinical medicine. The accuracy of the reagent used to determine blood group is not a quality metric in the abstract. It is a patient safety imperative. Eryclone Anti-A,B Monoclonal Antibody Reagent is a blood grouping reagent for the identification of A, B, and AB blood group antigens on human red blood cells, produced using monoclonal antibody technology. The reagent is produced from monoclonal antibodies with a defined, consistent specificity — unlike polyclonal antisera derived from human donors, which can show lot-to-lot variability in antibody titre and specificity. The Anti-A,B reagent detects both A and B antigens simultaneously, making it the cross-confirming reagent in the ABO typing panel. It is used in combination with Anti-A (blue) and Anti-B (yellow) reagents to provide the full ABO forward typing result. An Anti-A,B reagent that reacts positively confirms the presence of at least one of the A or B antigens — providing a particularly useful check on weak A or B subgroup expressions that might be missed by the individual anti-A or anti-B reagents alone. The reagent is formulated with inert protein additives and preservatives that maintain its potency and specificity throughout its shelf life. The dropper bottle format allows controlled, single-drop dispensing directly onto the tile or microplate well for slide, tile, or tube testing methods. For blood banks, transfusion laboratories, and hospital laboratories that perform ABO blood grouping, monoclonal blood grouping reagents are a consistently purchased item with regular replacement cycles tied to lot expiry. Indian-manufactured blood grouping reagents meeting CE or national certification standards offer competitive pricing relative to imported alternatives. Sara Wellness exports diagnostic reagents and laboratory consumables to medical distributors internationally.
Disposable ESR Pipette (Westergren Method)
The erythrocyte sedimentation rate is one of those tests that sits squarely at the intersection of clinical usefulness and analytical simplicity. No reagents. No analyser. No enzyme reactions. Blood, a pipette, a rack, and an hour. The rate at which red cells fall through plasma carries remarkably useful diagnostic information about the degree of acute-phase inflammatory response, infection burden, or tissue damage — and the Westergren method has been the reference standard for measuring it for over a century. Disposable ESR pipettes for the Westergren method are single-use plastic pipettes that replace the original glass Westergren tubes that were the backbone of ESR testing before single-use disposables became the standard. The disposable format eliminates the cleaning and cross-contamination risks associated with reusable glass tubes, which is a meaningful infection control advantage in high-volume clinical laboratories and blood bank settings. The Westergren method requires a pipette with a standardised internal bore diameter of 2.55mm and a scale length of 200mm graduated from 0 at the top to 200 at the bottom. Blood prepared at a 4:1 dilution with sodium citrate solution is drawn into the pipette to the 0 mark, the pipette is placed vertically in the ESR rack, and the column height of red cell sedimentation is read after exactly 60 minutes. The reading in millimetres per hour is the ESR value. Disposable ESR pipettes are manufactured from medical-grade clear plastic that provides the same visibility as glass for reading the sedimentation column endpoint. The pipette dimensions comply with ICSH reference Westergren method specifications. For laboratory supply distributors, disposable ESR pipettes are a high-volume consumable purchased by any laboratory performing manual ESR testing. Automated ESR analysers are increasingly common but manual Westergren testing remains widely used in resource-limited settings, smaller clinics, and as the reference method for quality checks. Sara Wellness exports laboratory consumables to medical distributors internationally.

ESR Blood Collection Tubes (Sodium Citrate, Westergren Method)
The ESR tube looks like any other vacuum blood collection tube from the outside. But its contents are specific to a single application, and using the wrong tube for an ESR test is one of those pre-analytical errors that gives you a number that means nothing. The sodium citrate concentration, the dilution ratio, the vacuum — all of it is calibrated precisely for the Westergren method. ESR blood collection tubes (black cap, sodium citrate) are purpose-made vacuum tubes for erythrocyte sedimentation rate testing using the Westergren method. The tube contains 3.8% sodium citrate anticoagulant, pre-measured to give a 1:4 citrate-to-blood dilution ratio when blood fills to the nominal draw volume. The pre-calculated vacuum draws exactly the correct amount of blood (typically 1.28ml into a 1.6ml sodium citrate volume, giving a 1:4 mix ready for direct pipetting), eliminating the manual dilution step that was required with older glass tube ESR systems. The black cap is the internationally standardised colour code that identifies the ESR tube in multi-tube phlebotomy draws, reducing the risk of mistaking an ESR tube for a citrate coagulation tube (light blue) in a busy venepuncture setting. For laboratories using closed-tube ESR systems (automated Westergren or Micro-ESR methods), the ESR tube feeds directly into the analyser's sampling system. For manual Westergren testing, the diluted blood from the ESR tube is aspirated into the Westergren pipette. For laboratory supply distributors, ESR tubes are ordered alongside EDTA tubes and citrate tubes as part of the complete phlebotomy tube range. Hospitals performing any haematological work order them in consistent volumes. Sara Wellness exports phlebotomy consumables including ESR tubes to medical distributors internationally.

Sodium Fluoride / Potassium Oxalate Vacuum Blood Collection Tubes (Grey Cap)
Blood glucose measurement sounds simple. Draw blood, measure glucose, record the number. But the number is only as accurate as the state of the sample at the time of analysis — and blood glucose is not a stable measurement in whole blood. Red blood cells and white blood cells continue to metabolise glucose through glycolysis after collection, consuming approximately 7-10 mg/dL per hour at room temperature. A sample drawn at 8 AM and analysed at 11 AM without proper preservation has potentially lost 20-30 mg/dL of its actual glucose content, turning what might have been a diagnostic finding into a normal result. Sodium fluoride / potassium oxalate vacuum blood collection tubes — the grey-capped tubes in the vacuum collection system — exist to prevent exactly this. Sodium fluoride is a glycolysis inhibitor that blocks the enzyme enolase in the glycolytic pathway, effectively stopping red blood cell glucose consumption. Potassium oxalate is an anticoagulant — calcium chelator — that prevents clot formation, keeping the blood in its liquid state for analysis. Together, these two additives preserve the blood glucose level in the sample within ±2 mg/dL of the true value for up to 24-48 hours at room temperature. The grey cap is the internationally standardised colour code for sodium fluoride tubes across all major vacuum collection systems, enabling rapid tube identification in clinical settings where multiple tube types are in use simultaneously. For clinical chemistry laboratories, point-of-care glucose testing, and any setting where fasting or postprandial glucose measurement is a clinical priority, grey-capped NaF/KO tubes are the appropriate pre-analytical tool. They are also used for lactate measurement and some other metabolite assays where anaerobic conditions or enzyme inhibition is needed to preserve analyte stability. For laboratory supply distributors, NaF tubes are ordered alongside EDTA and citrate tubes as part of the complete phlebotomy tube portfolio. Sara Wellness exports laboratory consumables to medical distributors internationally.

HIV Tri-Dot Test Kit by J. Mitra
Some clinical tools look simple and are. The HIV Tri-Dot is one of them. A dot-format rapid immunoassay, three antigen dots on a nitrocellulose membrane, capable of detecting HIV-1 and HIV-2 antibodies from a fingerstick blood sample in under 20 minutes, with visual result reading requiring no instruments and no specialized training. J. Mitra's HIV Tri-Dot is one of India's longest-established rapid HIV tests. The three-dot configuration refers to three antigen spots on the membrane: the first contains HIV-1 specific antigen, the second HIV-2 specific antigen, and the third is a procedural control. A coloured dot at any antigen position indicates a reactive result. Presence of the control dot confirms test validity. The format has proven particularly well-suited to voluntary counselling and testing centres, antenatal care programs, mobile outreach settings, and blood bank pre-transfusion emergency screening. It is listed in several national HIV testing algorithm recommendations and has been evaluated in field conditions with validated performance data. Manufactured by J. Mitra and Company under CDSCO license, the Tri-Dot meets the quality requirements for government and private health program use. For procurement buyers serving national AIDS control programs, blood banks, and HIV testing organizations, the J. Mitra Tri-Dot is a brand with a documented institutional procurement track record. Sara Wellness exports J. Mitra diagnostic kits and HIV testing products from India for international importers and public health procurement offices. Fifteen years of export experience, flexible minimum orders.

J. Mitra Hepacard
HBsAg rapid testing changed what HBV screening could reach. A test that needs no power, no washer, no microplate reader, and no laboratory technician can go to a primary health centre in any district. ELISA cannot. That geography gap is where the Hepacard earns its place. J. Mitra Hepacard is a one-step immunochromatographic rapid test for the qualitative detection of Hepatitis B Surface Antigen in human serum, plasma, or whole blood. The card format uses a nitrocellulose membrane coated with HBsAg-specific antibodies at test and control line positions. HBsAg in the sample binds to colloidal gold-conjugated detector antibody, and the complex migrates up the membrane to produce a visible coloured band at the test line within 15-20 minutes. Two lines means reactive. One line at the control position means non-reactive. No control band means the test is invalid regardless of the test line. That is the reading logic, and it is the same across the immunochromatographic test category globally. Hepacard is used in blood banks for donor screening, antenatal programs for maternal HBV status, pre-operative screening, community surveillance, and outpatient settings where access to ELISA infrastructure is limited or unavailable. J. Mitra is one of India's largest IVD manufacturers with CDSCO regulatory approvals and international export certifications. Sara Wellness has been exporting J. Mitra diagnostic products from India for 15 years with full export documentation for international buyers.

J Mitra & Co. 3rd Generation HCV Microlisa Test Kit
HCV screening is a public health priority that has been seriously underestimated in resource-limited markets. The WHO estimates roughly half of people living with hepatitis C don't know it. Blood banks, antenatal clinics, dialysis units, and population screening programs all need reliable, scalable, cost-effective HCV detection that doesn't require sophisticated infrastructure. J. Mitra's 3rd Generation HCV Microlisa is an ELISA for the qualitative detection of antibodies to Hepatitis C Virus in human serum and plasma. Third-generation means it detects antibodies against HCV core, NS3, NS4, and NS5 antigens. That broader coverage closes the diagnostic window period compared to second-generation assays, reducing the risk of window-period false negatives in recently infected donors. The 96-well microplate format makes this test economical for batch processing. One kit, one run, 96 samples. That throughput aligns with the workflow of mid-size diagnostic laboratories handling blood bank screening, pre-operative panels, and outpatient diagnostics simultaneously. J. Mitra & Co. is one of India's most established in-vitro diagnostics manufacturers, with regulatory approvals and export certifications for multiple international markets. Their ELISA platforms are used across Asia, Africa, and the Middle East by government hospitals, private laboratory chains, and blood banks. Sara Wellness has been exporting J. Mitra diagnostic kits and laboratory reagents from India for 15 years. Competitive pricing and full documentation for customs clearance are standard.

Meril MERISCREEN HIV 1-2 WB
HIV testing in high-burden settings moves fastest when the test does not move slowly. ELISA workflows, with their multi-hour turnaround and instrument dependencies, are the right choice for high-volume blood bank screening. For clinical diagnosis, counselling-supported testing, outreach programs, and emergency screening, what is needed is a result in 20 minutes that requires no laboratory infrastructure whatsoever. Meril MERISCREEN HIV 1-2 WB is a rapid immunochromatographic test for the simultaneous detection of antibodies to HIV-1 and HIV-2 in whole blood, serum, or plasma. The whole blood capability is clinically significant. It means the test can be performed directly from a fingerstick sample without centrifugation or processing, making it genuinely portable and suitable for field use. The test uses a nitrocellulose strip with separate capture zones for HIV-1 and HIV-2 antibodies. A colloidal gold-labelled antigen conjugate migrates with the sample. Antibodies in the sample bind the conjugate and are captured at the respective zone, producing a visible red line. A control line confirms test validity. Results are read at 20 minutes without any instrument. Meril Diagnostics is one of India's established in-vitro diagnostic manufacturers, with CDSCO approval and distribution across government health programs, blood banks, and clinical labs. For procurement buyers supplying HIV testing programs, blood banks, antenatal clinics, voluntary counselling and testing centres, and emergency departments, MERISCREEN is a recognized name with institutional supply experience. Sara Wellness exports Meril diagnostic products and rapid test kits from India with 15 years of verified export history and direct relationships with international health program procurement offices.

Hindustan Syringes Vaku-8 Blood Collection Set
Venipuncture is one of the most performed clinical procedures in medicine, performed millions of times daily in hospitals, clinics, and community health centers worldwide. The tools used for it directly affect patient comfort, sample quality, and hemolysis rates. That is why blood collection set selection matters far beyond its apparent simplicity. The Hindustan Syringes Vaku-8 Blood Collection Set is a winged infusion set (butterfly needle) designed for venipuncture in standard blood collection tube systems. The set consists of a winged needle with an attached flexible PVC tube and a needle adapter at the distal end that connects to vacuum collection tube holders. The winged design allows secure fingertip grip during insertion and stable positioning during sample collection. The Vaku-8 collection set is compatible with standard vacuum collection tube systems using Vacutainer or similar holder formats. Once the needle is inserted and the tube holder is attached, the vacuum in the collection tube draws blood through the tubing automatically and at a controlled rate. The multi-draw adapter allows multiple tubes to be collected from a single venipuncture. The flexible tubing between the needle and adapter reduces needle movement during tube changes, which is a genuine patient comfort benefit. It allows the needle to remain still in the vein while the clinician manipulates the tube holder and changes tubes. For blood banks, clinical laboratories, hospital phlebotomy services, and outpatient diagnostic centers, butterfly blood collection sets are a consumable with demand driven by collection volume rather than patient volume. Sara Wellness exports Hindustan Syringes phlebotomy products and laboratory consumables from India with 15 years of supply experience and institutional pricing for importers worldwide.

Hitachi Sample Cups 500 Pack
Chemistry analyser consumables are where laboratory efficiency quietly lives or dies. The analyser is visible. The sample cups, the reagents, the tubes, these flow through the laboratory invisibly until they don't. A delayed reagent shipment or an incompatible cup specification brings the workflow to a halt in ways that are immediately and expensively noticed. Hitachi sample cups are precision-manufactured polystyrene or polypropylene cups designed for use in Hitachi automated clinical chemistry analysers. These cups serve as the primary sample vessel from which the analyser aspirates patient serum or plasma for analysis. The cup geometry, material, and optical properties are specified by Hitachi to ensure compatibility with the aspirating probe dimensions, the cup identification system (where applicable), and the analyser's cup-handling mechanism. The 500-cup pack format reflects typical laboratory throughput for mid-volume chemistry labs that run 200-500 tests per day. The cups are individually designed with a flat base for stable positioning in the analyser tray, a precisely dimensioned inner diameter for accurate probe insertion depth, and materials that do not interact with the chemical reagents in the analysis. For laboratory supply managers, hospital procurement teams, and diagnostic network supply coordinators, Hitachi sample cups are a category where substitution with generic cups carries real analytical risk. Probe damage, liquid level sensing errors, and aspiration failures are documented consequences of cup-analyser incompatibility. Sara Wellness has been exporting laboratory consumables and analyser accessories from India for 15 years. We supply buyers across Africa, Southeast Asia, and the Middle East with compatible laboratory consumables at competitive institutional pricing.

Yellow Gel Clot Activator Tubes 4ml
In a diagnostic laboratory, blood collection tubes are not glamorous. But the wrong tube at the wrong moment produces results that are either meaningless or actively misleading, and that costs time, sample, and sometimes patient welfare. Getting the tube right is basic. Getting it right at volume is what laboratory procurement is about. Yellow gel clot activator tubes are serum separator tubes (SSTs), the workhorse of clinical chemistry. The 4mL format is one of the most commonly requested volumes for adult serum collections in outpatient and hospital laboratories. The tube contains a clot activator coating on the interior wall that triggers and accelerates fibrin formation after venipuncture. The gel separator creates a stable barrier between serum and cellular components during centrifugation, preventing re-mixing and preserving sample integrity for transport and testing. The gold or yellow colour coding is universally recognized across most laboratory collection systems as the indicator for serum separation. Phlebotomists, nurses, and laboratory staff work with these tubes daily by reflex. Getting a consistent, correctly manufactured tube matters for workflow reliability. These 4mL tubes are compatible with standard 16mm draw tube holders and most automated laboratory analysers. The vacuum draw is calibrated to collect the correct blood volume for the gel-to-blood ratio required for effective serum separation. After collection and inversion for clot activation, the tubes are centrifuged, and the serum layer above the gel barrier is used for chemistry, immunology, serology, and other serum-based assays. For laboratory consumables importers, institutional hospital procurement offices, and diagnostic chain supply managers, yellow gel clot activator tubes represent one of the highest volume reorder categories in laboratory supplies. Sara Wellness exports laboratory consumables and blood collection tubes from India for over 15 years. Bulk pricing, flexible MOQs, and export-ready documentation for international buyers.
HIV Rapid Test Kit 2 Test
HIV diagnosis in modern clinical practice requires at least two independent reactive results before a positive diagnosis can be confirmed. One rapid test, however sensitive, cannot stand alone as the basis for a life-altering diagnosis. The second test in the algorithm exists to protect patients from false positives, and the choice of which test to use in that position matters. The HIV Rapid Test Kit 2 Test is an immunochromatographic rapid test for the detection of antibodies to HIV-1 and HIV-2, formulated to serve as the second-line confirmatory test in a two-test or three-test HIV testing algorithm. For an algorithm to be statistically valid, the second test must use a different antigen preparation or different assay format than the first, so that a cross-reactive false positive on test one is not confirmed by test two. The kit detects both HIV-1 and HIV-2 antibodies in serum, plasma, or fingerstick whole blood within 20 minutes without laboratory instrumentation. Results are read visually. The test design incorporates a separate control line to confirm test validity. For procurement buyers managing HIV testing programs, national AIDS control inventory, blood bank screening supply, and antenatal testing services, pairing two different branded rapid tests from different manufacturers with different antigen compositions is standard practice. Buyers supplying complete two-test algorithm kits to health facilities need both components consistently available. Sara Wellness exports HIV rapid test kits and diagnostic supplies from India with 15 years of verified supply experience for health programs across multiple international markets.

HBSAG Elisa Hepalisa 96 Test Pack
When rapid tests are not enough and volume matters, ELISA is the answer. Blood banks processing hundreds of donations per week need throughput and sensitivity that individual rapid cards cannot provide. ELISA platforms are how national blood transfusion programs, large private blood banks, and hospital transfusion services operate at scale with defensible analytical performance. Hepalisa is J. Mitra & Co.'s ELISA kit for HBsAg detection, formulated for 96-well microplate batch processing. The sandwich ELISA format uses anti-HBsAg antibodies coated on the plate, captures HBsAg from the patient sample, detects it with an enzyme-conjugated antibody, and measures the resulting colour development spectrophotometrically. The sandwich format offers high specificity relative to competitive formats. ELISA sensitivity is significantly greater than immunochromatographic rapid tests, which matters for window-period infections and low-level antigenaemia. Blood bank regulatory standards in most countries specify minimum sensitivity thresholds that Hepalisa is validated to meet. For diagnostic laboratories and blood banks processing high volumes, the 96-well kit format fits standard workflow. It requires a microplate reader, washer, and incubator, which are common in mid-size and larger laboratories. Sara Wellness has been exporting J. Mitra ELISA diagnostic kits from India for 15 years. We supply blood banks, hospital laboratories, and diagnostic chains across Africa, the Middle East, and Southeast Asia with reliable supply and full export documentation.

Haemoglobin (HB) Tubes
Point-of-care haemoglobin measurement in resource-limited settings still relies, in many parts of the world, on the Sahli method and the calibrated haemoglobin tube. Automated haematology analysers have displaced it in well-resourced laboratories. They have not displaced it in field settings, community health programs, antenatal clinics without reliable electricity, and rural health posts where what you have is a trained health worker, a calibrated glass tube, and a colour comparator. Haemoglobin tubes for the Sahli method are graduated glass tubes calibrated in g/dL. Blood is collected to the specified volume mark, mixed with 0.1N hydrochloric acid to convert haemoglobin to acid haematin, diluted to match the built-in reference standard in the haemoglobinometer holder, and the tube graduation is read as the haemoglobin concentration. For national anaemia screening programs, maternal health platforms conducting iron supplementation tracking, school health teams, and military medical units, the Sahli tube remains a practical, validated tool that does not depend on power, refrigeration, or sophisticated reagent handling. Procurement buyers for public health programs and institutional laboratory supply chains will find these tubes available in pack sizes suited for high-volume ordering. Reorder cycles are tied to program scale and testing frequency rather than individual clinical throughput. Sara Wellness has been exporting laboratory consumables and public health diagnostic supplies from India for 15 years. We serve institutional importers across Africa, the Middle East, and South Asia with competitive pricing and reliable export documentation.