
3M Coban Self Adherent Wrap
3M Coban Self-Adherent Wrap | The Bandage That Holds Itself Together (And Your Dressings Too). No pins. No clips. No tape. No sticky residue pulling at skin when it comes off. If you have ever watched a nurse spend three minutes trying to fasten a conventional bandage around a patient's fingers or toes and thought, "there has to be a better way" — Coban is that better way, and it has been for decades. 3M Coban Self-Adherent Wrap is a cohesive elastic bandage that sticks only to itself. Not to skin. Not to hair. Not to clothing or drapes. It is lightweight, conformable, and breathable, designed for clinical applications including blood draws, dressing securement, immobilization, and secondary securement for difficult-to-dress areas like the head, toes, and fingers. Once applied, it stays put without loosening, which matters considerably when you are wrapping an active patient or dressing a joint that moves. The wrap can be worn for up to 7 days on the leg when used as part of a venous leg ulcer management program.It conforms to body contours, adjusts during application, and cuts easily with scissors (or tears by hand in the non-latex version). That versatility is a large part of why this product has found a home in everything from post-blood-draw coverage to sports injury support to veterinary care. Available in five widths: 1" x 5 yds, 2" x 5 yds, 3" x 5 yds, 4" x 5 yds, and 6" x 5 yds. (Sky Dental Supply) The image shows the 10 cm (approximately 4") skintone variant, which is the most widely exported size globally for hospital and clinic wound care applications. Color options include Tan, Blue, Red, Neon, and Rainbow assorted packs, making it easy to use color-coding for patient differentiation or departmental organization. Both latex and non-latex (Coban NL) variants are available, with the non-latex version featuring a hand-tear scrim structure that eliminates the need for scissors — a practical advantage in fast-paced clinical environments. Wholesale pack configurations run from individual rolls to cases of 6 packs with 5 rolls each (30 rolls total) for the 1" size, scaling up to case packs of 12 to 36 rolls depending on the width selected. Distributors supplying hospital systems or clinic chains will find the case-pack format straightforward to inventory and reorder. Here is the supplier conversation that matters. Sara Wellness has been exporting wound care products and medical supplies from India for 15 years. We know that cohesive bandages like Coban are high-turnover, repeat-order products for distributors. The reason buyers keep coming back to us is not a great sales pitch. It is because the orders arrive correctly, the documentation is clean, and when something needs sorting, there is a real person picking up the phone to sort it. We work one-on-one with every importer. MOQs are negotiable depending on your market and order frequency. Pricing is competitive. And for buyers interested in private-label cohesive bandage alternatives under their own brand, we have the contract manufacturing infrastructure to make that happen. Each shipment comes fully documented for customs clearance, properly packed for international freight, and with the specifications confirmed before it ships. No surprises at the port. If you are distributing wound care products and need a reliable, consistent supply of self-adherent cohesive wraps, Sara Wellness is worth a conversation. Available Sizes: 1", 2", 3", 4", 6" widths (all x 5 yds). Colors: Tan/Skintone, Blue, Red, Neon, Assorted. Variants: Latex and Latex-Free (Non-Latex). Pack sizes: 5-roll packs to cases of 30-36 rolls.

3M Crepe Bandage
3M Crepe Bandage | The Workhorse of Every Well-Stocked Medical Supply Cabinet. Some products are called "essential" so often that the word stops meaning anything. Then you try running a busy ward, a sports injury clinic, or an outpatient department without crepe bandages for two days, and the word suddenly makes complete sense again. The 3M Crepe Bandage is one of those products. Quietly indispensable. Used thousands of times a day across hospitals, clinics, physiotherapy centers, and first aid stations worldwide, and almost never discussed until the shelf runs out. Distributors who supply healthcare facilities know the type — fast-moving, repeat-order, and completely non-negotiable from a stocking standpoint. Made from cotton with a high elastic backing and fast edges, this bandage is manufactured in accordance with British Pharmacopeia specifications. The fully stretchable material applies compression to offer comfort when worn throughout the day. That BP compliance is not a minor detail — it is what makes this bandage acceptable to procurement departments in regulated markets across Europe, the GCC, Southeast Asia, and beyond. The bandage is 100% cotton and latex-free, which matters considerably when supplying facilities that follow strict allergy and infection control protocols. Latex-free certification has quietly become a default expectation in many international hospital procurement specs, and this bandage meets that requirement without any special ordering. Available in three standard widths: 8 cm x 4 m, 10 cm x 4 m, and 15 cm x 4 m, covering everything from limb support and ankle wrapping to broader wound dressing securement and post-operative bandaging. The 10 cm size is the most exported globally for general hospital use. The 15 cm is commonly used in orthopedic and post-surgical applications where broader coverage is needed. Typical applications include sprains, strains, dislocations, painful joints, post-operative conditions, sports injuries, and general surgical and orthopedic support bandaging. For importers, the numbers that matter most: single rolls for retail clinic markets, boxes of 10 rolls per size for mid-level wholesale and institutional supply, and full export cartons of multiple boxes for large-volume orders. These configurations make it straightforward to stock across different market segments without overcommitting to any single format. Now, the part of the conversation that separates one supplier from the next. Sara Wellness has been exporting medical consumables from India for 15 years. Crepe bandages fall squarely into the category of products we know inside out: high turnover, tight specs, and buyers who do not tolerate surprises. We ship correctly the first time. Documentation is clean. Packing is export-grade. And if something does need sorting, you reach a person who can actually sort it. We work one-on-one with every buyer. MOQs flex to fit your market and order cycle. For buyers interested in a private-label version or a contract-manufactured equivalent under their own brand, we have the manufacturing infrastructure to make that work too. India produces BP-compliant cotton crepe bandages at price points that consistently undercut many other sourcing regions without cutting corners on quality. That is the sourcing advantage we offer, plainly stated. Available Sizes: 6 cm x 4 m, 8 cm x 4 m, 10 cm x 4 m, 15 cm x 4 m. Pack options: Single roll, Box of 10 rolls, Box of 12 rolls, Export cartons (multiple boxes per size). Latex-free. BP-compliant.

3M Elastic Adhesive Bandage
Post-operative dressing securement is one of those tasks that looks straightforward on paper and then turns complicated the moment you try securing a bulky abdominal drain or an irregularly contoured shoulder dressing. Standard tapes slide. Cotton bandages lose tension. And nobody has time to rewrap the same dressing three times in one shift. The 3M Elastic Adhesive Bandage was designed to solve exactly that problem without requiring extra steps or secondary fasteners. This is a highly elastic, adhesive-backed bandage engineered for post-operative wound dressing securement, compression management, and fixation of medical devices in challenging anatomical sites. The elastic backing stretches and conforms to body contours, maintaining secure adhesion even over joints, curved surfaces, and areas subject to movement. That conformability matters considerably when dressing sites like shoulders, elbows, knees, and irregular abdominal contours where rigid bandages fail. The adhesive coating provides a high degree of adhesion strong enough to secure heavy or bulky dressings, drainage tubes, and medical devices without additional tape or clips. The bandage features fast edges designed to prevent unraveling and maintain structural integrity throughout wear time. Unlike non-adhesive elastic bandages that require pins or clips, this bandage adheres directly to itself and to skin, staying in place through patient movement and daily activities. The elastic material applies controlled compression to manage edema and swelling while accommodating body movement without restricting circulation. It stretches according to swelling or motion, reducing the risk of mechanical injury to skin from over-tightening. This makes it particularly useful in post-surgical settings where swelling fluctuates during the initial recovery period. Clinically, this bandage is used in post-operative care for securing wound dressings, in orthopedic applications for compression and support, and in general wound care for fixation of dressings over irregular body contours. It is widely used in surgical wards, recovery rooms, orthopedic clinics, and home healthcare settings. Available in widths ranging from 8 cm to 15 cm and lengths from 1.5 m to 6 m, the bandage accommodates different dressing sizes and clinical applications. The 10 cm x 4 m size is the most commonly stocked for general post-operative use. The 15 cm width is preferred for larger dressings and broader compression applications. For distributors supplying hospitals, surgical centers, and home healthcare providers, elastic adhesive bandages are high-turnover consumables with predictable reorder cycles. They are essential inventory for any facility managing post-operative patients or wound care cases. Sara Wellness has been exporting wound care products and surgical supplies from India for 15 years. Elastic adhesive bandages are products where adhesive performance and material elasticity directly impact clinical outcomes. We source from manufacturers whose production standards meet international specifications for medical-grade adhesives and elastic materials. We work one-on-one with every buyer. MOQs are flexible because we know that a distributor testing a new wound care line is in a different position from an established wholesaler managing regular hospital supply contracts. For buyers interested in contract manufacturing or private-label versions of equivalent elastic adhesive bandages, we have the infrastructure to support that. Each shipment includes complete export documentation: commercial invoices, packing lists, certificates of origin, and material safety data sheets where required for customs clearance.

3M Electrosurgical Patient Return Electrode Plate
The difference between a routine electrosurgical procedure and one with complications can come down to something as mundane as whether the return electrode maintained proper skin contact for the entire case. Nobody thinks about the grounding pad until it fails. Then suddenly everyone is thinking about it. The 3M Electrosurgical Patient Return Electrode Plate is engineered specifically to be the piece of equipment that nobody has to think about twice during surgery. This is a disposable, single-use dispersive return electrode designed to safely return electrosurgical energy from the patient's body back to the electrosurgical generator. The pad provides a large conductive surface area that disperses current over a broad contact zone, minimizing heat concentration and reducing the risk of patient burns at the electrode site. That broad dispersion is what separates a properly designed return electrode from the kind that causes thermal injuries. The conductive adhesive is water-based hydrophilic gel that flows uniformly into skin crevices and irregularities, maximizing skin-to-pad contact and ensuring reliable electrical conductivity throughout the procedure. The hydrogel formulation provides strong adhesion without leaving residue on the skin after removal, which matters considerably when dealing with friable or sensitive patient skin. The non-conductive border adhesive surrounds the conductive area to maintain electrical isolation and prevent edge burns. Split (dual foil) electrode designs are compatible with electrosurgical generators equipped with Contact Quality Monitoring Systems (CQMS), including all major monitoring platforms like REM, ARM, NESSY, and PSS. The split design allows the generator to continuously monitor pad-to-patient contact quality and alert surgical staff immediately if impedance rises or contact is compromised. This active monitoring significantly reduces the risk of alternate-site burns, which can occur when the return path is inadequate. The foam backing material is lightweight, conformable, and biocompatible, tested negative for cytotoxicity, skin sensitization, and acute cutaneous irritation. Scalloped edges improve adherence and reduce lifting at pad borders. The pad is designed for universal patient use where full skin contact and a suitable placement site can be obtained, accommodating both adult and pediatric applications depending on size selected. Clinically, these electrodes are used in any surgical procedure involving electrosurgical cutting or coagulation. Common applications include general surgery, gynecological procedures, orthopedic surgery, ENT procedures, and plastic surgery where monopolar electrosurgery is employed. For distributors supplying operating rooms, ambulatory surgery centers, and hospital surgical departments, electrosurgical return electrodes are high-turnover disposable supplies with predictable consumption rates tied directly to surgical volume. They are essential inventory for any facility performing electrosurgery. Sara Wellness has been exporting surgical consumables and operating room supplies from India for 15 years. Electrosurgical return electrodes are safety-critical devices where material quality, adhesive performance, and conductive gel composition directly impact patient outcomes. We source from manufacturers whose production standards meet international specifications for medical-grade materials and electrosurgical safety. We work one-on-one with every buyer. MOQs are flexible because we know that a distributor adding a surgical consumables line is in a different position from an established wholesaler managing regular hospital supply contracts.
Trupace Cardiac Pacing Wire
Open-heart surgery is already the most consequential thing a surgical team will do on any given day. Everything that goes into that procedure — every wire, every suture, every disposable — carries the same weight of consequence, even if the price tag doesn't reflect it. The temporary pacing wire is one of those quiet essentials. It keeps the heart beating to the right rhythm in the hours immediately after surgery, and then it is removed. Simple, critical, unremarkable to everyone except the patient. Trupace, made by Sutures India, is a temporary cardiac pacing wire built for exactly this role. The conductor is a braided stainless steel multifilament at 0.3mm gauge, insulated along most of its length with nylon — specifically polyamide 6, a coating chosen for its consistent electrical insulation properties and biocompatibility. The distal un-insulated end is crimped to a curved needle for fixation to the myocardium. The proximal end connects via a straight breakpoint needle that exits through the chest wall and connects to the external pacemaker. The breakpoint needle design allows clean removal without requiring wire cutters on the skin-exit end. The 60cm working length covers the standard anatomical requirements for both atrial and ventricular placement during thoracic procedures. Cardiac surgeons performing CABG, valve repair, or any open-heart case where temporary epicardial pacing may be needed will recognise this configuration immediately. For surgical supply importers, hospital procurement managers, and regional distributors who source cardiac surgical consumables, Trupace comes from a credible, established Indian manufacturer. Sutures India has been producing surgical materials for decades and supplies into both the domestic hospital market and international export channels. The product arrives in boxes of 12, individually sterile packed, ethylene oxide sterilised, with the shelf life and documentation expected for cardiac surgical use. Sara Wellness supplies Trupace and other Sutures India surgical consumables to buyers across Africa, the Middle East, and Southeast Asia. Cardiac surgical supply chains in these regions need a supplier who understands clinical product requirements — not just logistics. We know the difference between a pacing wire and a suture even if neither of us is in the operating theatre, and we know which documentation your market's regulatory authority needs before the product can move. Pricing is quoted on volume. MOQs are flexible for buyers combining multiple cardiac surgical SKUs in one order. If you're building or restocking a cardiac surgery consumable line for a hospital group or distributor network, we're a straightforward conversation away from a quote.

Commode Chair (Standard)
Patient mobility after surgery, injury, or illness does not follow a fixed timeline. For some it is days, for others months. During that period, the activities that healthy people do without a second thought — using the toilet, washing, moving between rooms — become significant physical challenges. The commode chair exists to address one of those challenges directly. A commode chair is a portable toilet seat mounted on a stable frame, designed to be placed at the bedside, in a bathroom, or wherever the patient needs it. It eliminates the distance and physical effort required to reach a fixed toilet, which matters enormously for patients with limited strength, balance problems, post-operative restrictions, or mobility impairment. Standard commode chairs feature a steel or aluminium frame with rubber-tipped legs for floor grip and stability, a comfortable seat with a central opening over a removable bucket, armrests for safe lowering and rising, and a lid that closes for hygiene between uses. The bucket lifts out cleanly for emptying and cleaning. Height adjustment on most models accommodates different patient heights and different transfer approaches. The design is deliberately straightforward. Complicated equipment requires training, maintenance, and sometimes courage to use. A commode chair that is simple and stable gets used consistently, which is what matters most for patient safety and dignity. For medical supply distributors serving home care supply chains, rehabilitation equipment importers, and hospital procurement teams, commode chairs are a high-volume, steady-consumption category. They are ordered by hospitals, nursing homes, physiotherapy centres, and home care equipment rental companies. Sara Wellness exports rehabilitation and mobility aids including commode chairs to distributors internationally. Competitive pricing, clear specifications, and flexible order quantities are how we operate.

Commode Chair with Wheels
Two patients can have identical diagnoses and very different mobility situations. One can transfer from bed to a stationary commode independently. The other cannot. For the second patient, the commode chair with wheels changes the entire care picture. It comes to the patient instead of requiring the patient to go to it. The wheeled commode chair incorporates castor wheels on the frame legs, allowing it to be rolled across smooth and semi-smooth floor surfaces. This serves two distinct clinical purposes. First, it allows a carer or nurse to move the chair to the patient bedside for use without physical lifting. Second, it allows the patient themselves — if they have sufficient upper body strength and cognition — to move the chair independently, reducing dependence on staff for every toilet visit. Wheel locking is an important safety feature. The castors on a quality wheeled commode include a locking mechanism that secures the chair in position during use, preventing movement during the act of transferring weight onto the seat or rising from it. A commode that moves during a transfer is a falls risk, and falls in hospitalised or post-surgical patients can have serious consequences. Armrests on wheeled commode chairs are typically fixed or removable. Removable armrests allow lateral transfer — shifting the patient from bed to commode without requiring them to stand. This is important for patients who cannot bear full weight. For hospital procurement and rehabilitation equipment distributors, wheeled commode chairs occupy a higher clinical need tier than stationary models. Hospitals, nursing homes, and rehabilitation wards order them in meaningful quantities. Sara Wellness exports rehabilitation and nursing aid equipment internationally, with 15 years of experience servicing medical supply distributors who need clear product specifications, competitive pricing, and no surprises in the supply chain.

Datt Softswab Sterile Gauze Swabs with X-Ray Line — 4s
A retained surgical swab is one of the worst categories of preventable surgical error. It is not a headline event, it is a system failure. Standard protocols — counting swabs before and after, using radiopaque markers, conducting imaging when counts are uncertain — exist because human counting is fallible, particularly in long procedures and emergency situations where the count has been disrupted. The X-ray detectable gauze swab exists to be the fail-safe. Datt Softswab Sterile Gauze Swabs with X-Ray Line contain a barium sulphate-impregnated thread woven into the gauze fabric. This thread shows up clearly on plain radiographs, making any accidentally retained swab visible on a post-operative X-ray even when embedded in tissue or a body cavity. The 4-ply construction gives these swabs their combination of absorptive capacity and structural integrity. They absorb wound exudate and blood effectively while maintaining enough body to be handled cleanly, counted accurately, and removed intact. Non-sterile gauze that sheds fibres into a wound is a contamination problem. Softswab's construction and sterilisation process maintain the fabric's integrity under use conditions. The product is individually sterilised using gamma radiation or ETO, maintaining sterility until the point of opening. Each swab is ready for direct application to surgical wounds, for cavity packing, or for irrigation assistance without requiring any preparation step. For hospital procurement teams and surgical supply distributors, X-ray detectable swabs are not optional in operative settings where abdominal or cavity procedures are performed. They are a patient safety requirement in most institutional surgical protocols. Sara Wellness exports wound care and surgical gauze products to distributors internationally with the complete documentation required for medical textile imports.

Flemingo Hernia Belt
Not every hernia patient is a surgical candidate on the day of diagnosis. Age, comorbidity, patient preference, and a waiting list that has its own timeline all mean that a significant number of patients need a conservative management option that provides reduction support while they wait for or decide against surgery. The hernia belt is that option. Flemingo is an established Indian orthopaedic support brand with a consistent presence in the rehabilitation equipment market. The Flemingo Hernia Belt provides external mechanical support to the hernia site, using a firm pad over the hernia orifice backed by an adjustable belt system that maintains compression across the inguinal or umbilical region throughout daily activity. The pad size, pad positioning, and belt tension can be adjusted to match the individual patient's anatomy and the extent of the hernia. The belt construction combines a breathable fabric outer layer with a firm inner pad that applies targeted pressure to the hernia site. Breathability matters in a product worn against the skin for extended periods, particularly in warm climates. The closure system uses either velcro or a buckle depending on the model, and adjustment is straightforward enough that patients can manage it independently at home. Sizes run from small through double extra-large, covering paediatric through large adult requirements. Single support for unilateral hernias and double support for bilateral hernias are both available as separate SKUs. For rehabilitation and orthopaedic supply distributors, hernia belts are a consistent category. They are prescribed by general surgeons, recommended by physiotherapists, and sold through pharmacy and orthopaedic retail channels. Post-surgical use to provide additional support during recovery is also a significant application. Sara Wellness exports Flemingo orthopaedic supports including hernia belts to distributors in multiple export markets. We have 15 years of experience navigating the brand-specific documentation requirements for orthopaedic export and price for volume relationships.

Covidien Appose Disposable Skin Stapler
Wound closure after a surgical incision should be as fast and reliable as the procedure that preceded it. Placing sutures closes the wound but adds time and the potential for inconsistency. Skin stapling does the same job in less time, with consistent closure geometry that does not depend on knot tension or stitch spacing. For the long incisions of general surgery, orthopaedic surgery, and Caesarean section, the skin stapler has become a standard part of the closure routine. The Covidien Appose ULC is one of the most widely recognised disposable skin staplers in the market. The design is built around an easy-squeeze trigger that fires one staple per press, an ejector spring that automatically disengages the staple to facilitate instrument removal in any direction, and a clear view of the tissue being stapled through the transparent cartridge window. The slim body reduces the visual obstruction of the wound site during placement. Two staple sizes are available. Regular staples are 4.8mm wide with a 3.4mm leg length for standard wound closure. Wide staples are 6.5mm wide with a 4.1mm leg length for thicker skin or situations where wider tissue bridge coverage is preferred. Both are made from stainless steel. The stapler is single-use and sterile from the packaging. There is no activation step — remove from packaging, position, fire. Each unit comes pre-loaded with 35 staples, which is sufficient for the majority of surgical incision closures in a single unit. For surgical supply distributors and hospital procurement teams, Covidien Appose is a recognised brand that surgical departments request specifically. Box-of-12 purchasing is the standard institutional format. Sara Wellness exports Covidien surgical products to distributors internationally, with 15 years of experience in medical device export documentation and logistics.

Covidien Surgipro Flat Mesh — 15 x 15 cm
Hernia repair accounts for approximately 20 million procedures globally each year. That makes it one of the most common elective surgical procedures performed worldwide, and mesh-based tension-free repair has been the standard of care for most hernia types for the past three decades. The quality of the mesh implanted influences the rate of hernia recurrence, the risk of mesh-related complications, and the patient's long-term experience. Surgeons who perform hernia repair regularly develop preferences for specific mesh products, and those preferences drive procurement decisions in surgical departments. Covidien Surgipro Flat Mesh is a nonabsorbable polypropylene surgical mesh designed for both open and laparoscopic hernia repair. The polypropylene construction is permanently implanted — the body incorporates the mesh through tissue ingrowth over time, forming a permanent scaffold that reinforces the abdominal wall defect. The mesh is knitted in an open-weave structure that allows tissue ingrowth while maintaining the dimensional stability required for proper repair. The 15x15cm flat sheet format is the most commonly ordered size for inguinal hernia repair, providing sufficient material for proper overlap of the hernia defect plus anchoring margin without requiring intraoperative trimming in most standard cases. For ventral or incisional hernias where larger coverage is required, the 30x30cm format is available. Bi-directional elasticity in the mesh construction means it can be cut to any shape required without fraying or unravelling at the cut edges. This is a practical property that matters in the surgical field. For surgical supply distributors and hospital theatre managers, Surgipro is a recognised Covidien (Medtronic) brand with institutional supply credibility. Sara Wellness exports surgical mesh products internationally with complete regulatory documentation for medical device imports.

Dr. Sabharwal's Sterile Surgical Pads
Not all wound care products come with the fanfare of branded surgical systems. Some of the most consistently used consumables in any hospital or clinic are the quiet, reliable ones that do their job without requiring a seminar: sterile gauze, surgical pads, absorbent dressings. The surgical pad is exactly that. It shows up at wound closures, post-operative dressing changes, intra-operative field management, and a hundred other moments in clinical practice where absorption and sterility are the only two requirements. Dr. Sabharwal's Sterile Surgical Pads are an Indian-manufactured sterile absorbent wound pad designed for surgical and clinical wound management applications. The pad is constructed from highly absorbent cotton gauze or non-woven fabric with a soft outer face and an absorbent core, individually wrapped and sterilised for single-use application. The sterile packaging maintains sterility until the moment of opening, providing a clean-field wound care product for both operating theatre and general ward use. Surgical pads in this category are used for: post-operative wound coverage between dressing changes; absorbing exudate at wound sites during the immediate post-operative phase; intra-operative field management where a large absorbent surface is needed without fine gauze that may shed fibres; debridement preparation; and wound packing in cavity wounds where bulk absorbency is needed. For hospital procurement offices, surgical supply distributors, and clinic chains sourcing wound care consumables, Indian-manufactured sterile surgical pads offer a cost-effective alternative to imported brands in a product category where clinical performance is determined by raw material quality and manufacturing standards rather than brand equity. India's wound care manufacturing sector is well-established, with a significant number of ISO and CE-certified manufacturers supplying to export markets across Southeast Asia, the Middle East, and Africa. Sara Wellness exports sterile wound care consumables to medical distributors internationally, with 15 years of export experience, competitive pricing from Indian manufacturing, flexible minimum order quantities, and complete export documentation handled end to end.
Ethicon Ethibond Excel Polyester Suture W4846 (USP 5, V-55, 4x75cm, Green)
Cardiovascular surgery, orthopaedic repairs, fascial closure in abdominal procedures, ophthalmology, neurological tissue fixation — these are not situations that tolerate a suture that degrades, stretches, or loses its tensile strength after placement. The tissues being approximated need to remain approximated indefinitely. The suture is expected to maintain its mechanical properties for the life of the patient. Ethicon Ethibond Excel is a non-absorbable braided polyester suture — polyethylene terephthalate (PET) monofilament fibres woven into a braided multifilament structure and coated with polybutylate, a unique coating compound developed by Ethicon. Polybutylate is a semi-crystalline co-polymer that bonds to the polyester braid mechanically rather than as a surface film, providing a coating that does not flake off or degrade over time. The coating gives Ethibond Excel its characteristic smooth tissue passage and handling properties without the coating failure that can occur with wax or silicone coatings on other braided sutures. The W4846 designation identifies a specific needle-strand combination: USP size 5 (EP gauge 6) green braided suture, four 75cm strands per package, fitted with the V-55 needle — a large, heavy-gauge 1/2 circle taper-cut needle designed for tough, fibrous tissue that requires more cutting force than a standard taper-point would provide. The V-55 needle is commonly used in large orthopaedic applications, fascial repair, and tendon surgery. The green dyeing provides high visual contrast against tissue, which is a clinical priority in procedures where suture retrieval and knot identification must be rapid and unambiguous. The braided structure provides reliable knot security — critical in permanent sutures where post-operative knot slippage would compromise the repair. For surgical supply distributors, Ethibond Excel is a standard catalogue item in suture product ranges, particularly those serving orthopaedic, cardiovascular, and general surgery units. Sara Wellness exports Ethicon surgical sutures to medical distributors internationally.
Ethicon Bone Wax W810 (Sterile Haemostatic Bone Wax)
Every surgeon who has drilled into bone — whether in neurosurgery, orthopaedics, thoracic surgery, or even complex dental implant placements — knows the specific frustration of bleeding from the cancellous bone surface. It does not respond to electrocautery the way soft tissue bleeding does. You cannot clamp a trabecular space. Suture ligation is not an option. And the blood keeps welling up from a surface that, absent intervention, will continue to bleed throughout the procedure. Bone wax was invented in the 1880s by the neurosurgeon Victor Horsley specifically for this problem. The product Ethicon sells as Bone Wax today is, in its essential concept, the same solution: a malleable wax substance pressed into the cut bone surface that physically tamponades the bleeding vessels in the cancellous space. No biochemistry. No coagulation cascade activation. A physical seal. Ethicon Bone Wax W810 is a sterile mixture of beeswax, paraffin, and isopropyl palmitate. Beeswax provides the structural framework and the tackiness that allows the wax to adhere to the bone surface. Paraffin adjusts the hardness and melting point so that the wax is workable at room temperature but does not soften excessively in the wound. Isopropyl palmitate is the wax-softening agent that ensures the product is pliable enough to be moulded onto the bone surface without crumbling. W810 is supplied as individually wrapped 2.5g tablets in a box of twelve. At the required moment in the procedure, the surgeon takes a small piece of wax, warms it briefly between the fingers to increase pliability, and presses it firmly onto the bleeding bone surface. The wax is rubbed into the cancellous surface with the finger or a pledget, spreading into the trabeculae and occluding the vessels mechanically. An important clinical note: bone wax is not absorbed by the body. It will remain permanently in the wound unless surgically removed. It should not be used in applications where bone healing or fusion is the desired outcome, as the wax barrier inhibits osteoblast ingrowth and bone regeneration. Sara Wellness exports Ethicon surgical consumables to medical distributors internationally.
Ethicon ENSEAL G2 Tissue Sealer
Minimally invasive surgery lives or dies by two variables: access and haemostasis. Access is the trocar and port problem. Haemostasis during tissue dissection and vessel division is a different challenge entirely — one that became dramatically more controllable with the development of advanced energy devices that seal and divide in a single instrument pass without the spray and char of monopolar cautery. The Ethicon ENSEAL G2 Tissue Sealer is an advanced bipolar energy device for laparoscopic tissue sealing and division. The instrument uses Ethicon's PTC (Positive Temperature Coefficient) technology, which is a temperature self-regulation system built into the jaw itself. Standard bipolar devices deliver RF energy at a set power level regardless of tissue impedance changes during sealing — which can result in local overheating as tissue desiccates and impedance rises. The PTC material in the ENSEAL G2 jaw increases its electrical resistance as temperature increases, which automatically reduces the energy delivery when the tissue is adequately sealed and beginning to overheat, without requiring feedback from an external generator algorithm. The G2 articulating version adds 360-degree jaw articulation — the instrument tip can be oriented in any direction from the 5mm shaft, which is a meaningful ergonomic and access advantage in laparoscopic procedures where the approach angle dictates the force vectors available for tissue manipulation. This matters most in sigmoid colectomy, anterior resection, hysterectomy, and any procedure where the target tissue is in a deep pelvic location that a straight-shaft instrument cannot access efficiently. The ENSEAL G2 seals vessels up to 7mm in diameter reliably, which covers the vast majority of the vascular structures encountered in laparoscopic general surgery. The system is compatible with a dedicated Ethicon energy generator but is also compatible with some generators from other manufacturers — confirm compatibility with the specific generator in use before procurement. Sara Wellness exports Ethicon advanced energy surgical devices to medical distributors internationally.
Ethicon Endopath Dextrus Minimally Invasive Access System
The trocar insertion step is the access moment in laparoscopic surgery. Everything that follows depends on the port being in the right place and on the abdominal wall and peritoneum being entered without causing inadvertent injury to underlying viscera or blood vessels. The design of the access system determines how controlled and how safe this critical first step is. The Ethicon Endopath Dextrus Minimally Invasive Access System is a visual-entry or optical trocar system designed for laparoscopic port insertion. Unlike traditional blind-entry trocars that penetrate the abdominal wall through a pointed or shielded mechanism without direct visualisation of the layers being entered, the Dextrus system is designed to be used with a 0-degree laparoscope placed within the obturator cannula, allowing the surgeon to watch each fascial layer as it is separated and entered under direct vision. Visual entry is widely advocated as a safer approach to trocar insertion than blind entry techniques, particularly for the primary port placement, as it allows immediate visualisation of the abdominal wall layers and enables recognition of adhesions, bowel attached to the anterior abdominal wall, or vascular structures that would be injured by a blind entry. The system uses a dilating entry mechanism rather than a cutting blade — blunt dilating access to the peritoneal cavity spreads the tissue fibres rather than cutting them, which is associated with less bleeding at the port site and potentially lower risk of port-site hernia from trocar-site fascial defects. The retention system within the cannula holds the port securely against the abdominal wall during the procedure without requiring external skin fixation. For laparoscopic surgery supply distributors, the Endopath Dextrus is catalogued alongside the complete Ethicon laparoscopic port range, serving general surgery, gynaecology, and urology theatres. Sara Wellness exports Ethicon laparoscopic access devices to surgical supply distributors internationally.
Ethicon Ethilon Non-Absorbable Nylon Suture NW3336
Nylon sutures occupy a specific position in the suture inventory that is not easily replaced: strong, smooth, non-reactive, and available in fine enough diameters to close skin on the face with minimal scarring. They are the suture of choice for skin closure, where the material will be removed after healing, and where the primary requirements are tissue-friendly passage, minimal scarring, and reliable knot security for the duration of healing. Ethicon Ethilon is a monofilament nylon suture — a single continuous filament of long-chain aliphatic polyamide polymer (Nylon 6 and Nylon 6.6) that provides a smooth, low-drag tissue passage without the interstices of a braided structure that could harbour bacteria. The monofilament structure also provides spring-back memory (the suture tends to return to its original straight shape when removed from the packaging), which requires more throws per knot to achieve secure knotting but also means the suture does not wick fluids along its length. The NW3336 is a specific product code identifying: USP size 2-0 (EP 3), black monofilament, with a 45mm 3/8 circle reverse cutting needle, and a 70cm strand length. The reverse cutting needle has a cutting edge on the outer (convex) curve of the needle, which reduces the risk of suture pullout through tissue compared to conventional cutting needles (which cut toward the wound margin). The 45mm needle provides good tissue clearance for general wound closure applications where the tissue thickness requires a larger needle radius. Black dye provides high visual contrast against skin and tissue for easy suture identification during placement and at removal. The 70cm working length provides adequate material for medium-length wound closures without excess strand to manage. For surgical supply distributors, Ethilon is one of the most widely ordered non-absorbable suture products — a standard skin closure option in virtually every surgical tray configuration. Sara Wellness exports Ethicon sutures to distributors internationally.
Ethicon Ligaclip Multiple Clip Applier (MCA)
Vascular ligation during surgery comes down to a simple mechanical requirement: a device that closes a vessel completely, holds it closed permanently, and can be placed with one hand while the other hand manages retraction or exposure. The ligating clip was designed to meet this requirement faster than suture ligation and with less need for precise knot-tying in tight operative fields. Ethicon Ligaclip Multiple Clip Applier (MCA) is a preloaded clip applier for titanium ligating clips used in open surgical procedures. Each applier comes preloaded with 20 or 30 titanium clips (depending on the model), with an automatic clip-advance mechanism that moves the next clip into the jaw after each application — no reloading required during the procedure, and no separate clip cartridge to manage at the scrub table. The mechanism works with a single hand: the jaws are positioned around the vessel, the handles are fully squeezed, and the clip is formed into a U-shape that closes around the vessel at a geometry calibrated to achieve complete occlusion without cutting through the vessel wall. When the handles are completely released, the next clip automatically advances into position. The entire sequence takes about two seconds per clip application. A proprietary groove system on the Ligaclip titanium clips — with patented lateral and transverse grooves on the inner jaw faces — provides secure interlocking between the formed clip and the vessel, preventing clip migration or rotation post-placement. This is a specific engineered feature that addresses the vessel clip migration events that occur with simpler clip designs in the immediate post-operative period. The range covers small, medium, medium/large, and large clip sizes for different vessel calibres. The lockout feature prevents clip application when the clip size is inappropriate for the tissue being ligated, reducing the risk of inadequate occlusion from an undersized clip. Sara Wellness exports Ethicon laparoscopic and open surgical instruments to distributors internationally.
Ethicon Ligaclip Extra Ligating Clips — Open and Endo Single Clip Applier
Multiple-fire clip appliers are efficient for high-volume sequential ligation. But there is a different clinical situation — the single critical vessel, the carefully dissected hepatic duct, the cystic artery confirmed clean and clear — where what the surgeon needs is not 20 clips advancing automatically but a single, precisely positioned clip placed with complete deliberate control. This is where the single clip applier earns its place on the instrument tray. Ethicon Ligaclip Extra is the single-clip system in the Ethicon Ligaclip family, covering both open and laparoscopic applications with a multi-patient use applier handle and disposable clip cartridges. Each cartridge contains a stack of 10 clips; the surgeon loads a fresh cartridge onto the applier handle and applies clips one by one, with each application requiring a deliberate reload from the cartridge — providing the manual pause that encourages deliberate individual clip placement rather than rapid sequential firing. The Ligaclip Extra clips carry the same titanium construction and patented groove design as the Multiple Clip Applier range — lateral and transverse grooves on the inner jaw faces that interlocked with the formed clip, preventing rotation or migration after application. The titanium clips are MRI-compatible, radiopaque on fluoroscopy, and inert in tissue. The open surgery applier handles come in multiple shaft lengths (5.75", 7.5", 10.5") and tip angles (straight, angled, right-angled) to reach different anatomical locations from the open operative field. The laparoscopic versions come with fixed 5mm shaft diameters for passage through standard 5mm trocar ports. For surgical supply distributors, Ligaclip Extra sits alongside the Ligaclip MCA in the catalogue, covering the complementary single-application use cases where the MCA's auto-advance mechanism is not the right tool. Sara Wellness exports Ethicon surgical clip products internationally.
Ethicon MONOCRYL (Poliglecaprone 25) Suture Y494G
Wound closure after elective facial surgery, delicate subcuticular skin closure, ophthalmic procedures, and any case where the surgeon wants the suture to dissolve cleanly within a few weeks without inflammation — this is where MONOCRYL earns its position as one of the most prescribed synthetic absorbable sutures in the world. Ethicon MONOCRYL is a synthetic absorbable monofilament suture composed of a copolymer of glycolide and epsilon-caprolactone (poliglecaprone 25). The glycolide-caprolactone chemistry was specifically engineered to produce a material with a short absorption timeline and a very low tissue reactivity profile — properties that distinguish it from older absorbable monofilaments. The absorption profile is predictable: MONOCRYL retains approximately 50-60% of its original tensile strength at 7 days and approximately 20-30% at 14 days post-implantation, with complete mass absorption by hydrolysis occurring within 90-120 days. The short strength retention window makes MONOCRYL the ideal choice for skin closure and subcuticular closures where the wound only requires suture support for 7-10 days, after which the healed tissue provides its own strength. For procedures requiring prolonged tissue support, polydioxanone (PDS II) or polyglactin (Vicryl) are more appropriate. The Y494G designation identifies a specific product: size 4-0 (EP 1.5), undyed (not violet), single strand 18" (45cm), P-3 needle (13mm, 3/8 circle, precision reverse cutting). The undyed formulation is preferred for cosmetically sensitive skin closure where violet pigment visibility at the skin surface is undesirable. The P-3 needle is a small, precise needle suited for delicate tissue passage. For surgical suture distributors, MONOCRYL is a high-volume, consistently ordered product across surgical, obstetric, and aesthetic surgery segments. Sara Wellness exports Ethicon sutures internationally.
Ethicon PDS II Polydioxanone Suture NW9262
Abdominal wall closure after major laparotomy demands a suture that is still doing its job weeks after the operation, when the patient is coughing, breathing deeply in physiotherapy, and beginning to resume activity. Mass closure of the abdominal wall requires adequate tensile support for the full healing cycle — typically 4-6 weeks — because the risk of burst abdomen peaks at the point when the abdominal wound is under dynamic functional load but has not yet developed adequate collagen maturity. Ethicon PDS II is polydioxanone — a synthetic absorbable monofilament suture with one of the longest strength retention profiles among absorbable suture materials. PDS II retains approximately 70% of its original tensile strength at 2 weeks post-implantation and approximately 50% at 4 weeks, providing meaningful mechanical support through the entire primary wound healing phase. Complete absorption by hydrolysis occurs between 182 and 238 days — well past the point at which support is needed. The NW9262 product code identifies a large, heavy version for major fascial and abdominal wall closure: USP size 1 (EP 4), violet monofilament, 150cm strand (length sufficient for continuous mass closure of a midline laparotomy), fitted with a 50mm 1/2 circle round body heavy needle. The round body needle has a tapered point and no cutting edge — it pushes through tissue rather than cutting, which preserves tissue integrity and reduces the risk of suture pullout through the widened needle track that a cutting needle creates. The violet colour provides excellent visibility in deep wounds during continuous suture placement and facilitates identification at any subsequent procedure if the suture is still present before absorption is complete. PDS II is also used for fascial repair in hernia surgery when a delayed-absorbable suture is preferred over permanent suture, and in paediatric cardiovascular surgery where tissue growth at the suture site is expected. Sara Wellness exports Ethicon sutures to medical distributors internationally.
Ethicon PDS Plus Antibacterial Polydioxanone Suture PDP9262T
Surgical site infection is a problem that clinical practice has been trying to solve since surgery began. Despite every advance in antibiotic prophylaxis, operating theatre ventilation, skin preparation, and surgical technique, SSI rates in major abdominal surgery remain a consistent source of patient morbidity, prolonged hospitalisation, and healthcare cost. The triclosan-coated suture was developed specifically to add an additional layer of protection at the one place that antibiotics and environmental controls cannot reach: the surface of the implanted suture itself. Ethicon PDS Plus Antibacterial Suture is polydioxanone monofilament — the same material, same absorption profile, same tensile properties, and same handling characteristics as PDS II — with the addition of triclosan (Irgacare MP), a broad-spectrum bacteriostatic agent coated onto each strand. Triclosan inhibits fatty acid synthesis in bacteria, disrupting their ability to form and maintain cell membranes. When bacteria attempt to colonise the suture surface, they encounter an antibacterial concentration that prevents biofilm establishment. In vitro data from Ethicon and peer-reviewed publications demonstrate that PDS Plus maintains antibacterial activity against S. aureus, MRSA, S. epidermidis, MRSE, E. coli, and K. pneumoniae for 17-23 days post-implantation. Clinical trial data (meta-analysis of 31 RCTs with over 14,000 patients) shows approximately 30% reduction in SSI risk with triclosan-coated sutures compared to non-coated equivalents. The National Institute for Health and Care Excellence (NICE, UK) specifically recommends triclosan-coated sutures as part of the SSI prevention care bundle. PDP9262T is a loop suture configuration: two needles attached to the ends of a 150cm strand looped through a single anchor — a format used for certain abdominal wall closure techniques where the loop is threaded through the wound and both needles close the fascia from opposite ends simultaneously. The CTX needle is a large, heavy 1/2 circle taper-point needle suitable for major abdominal fascia. Sara Wellness exports Ethicon antibacterial sutures to surgical distributors internationally.
Ethicon PML01 Prolene Polypropylene Hernia Mesh
Hernia repair has one fundamental objective that every mesh manufacturer works toward: create a repair that does not recur, is tolerated by the patient's body, and does not cause complications that make the patient wish the hernia had been left alone. Getting all three right simultaneously is not straightforward. The mesh chemistry, the pore architecture, the weight, the bending stiffness, and the long-term integration profile all matter. Ethicon Prolene Polypropylene Mesh has a track record in hernia repair that is measured in decades. The material is the same knitted polypropylene monofilament used in PROLENE polypropylene suture — a proven, stable polymer that resists enzymatic degradation in tissue and retains its physical properties indefinitely after implantation. The knitted construction provides both the tensile and burst strength required for hernia repair and the bidirectional elastic property that allows the mesh to conform to the three-dimensional contours of the abdominal wall and accommodate the mechanical stresses of intra-abdominal pressure, coughing, and physical activity. The mesh is knitted in a pattern that permits cutting into any required shape without unravelling the edges — a practical advantage that allows the surgeon to trim the mesh to the exact dimensions of the defect or overlap required, particularly for irregular defects or unusual repair geometries. The cut edges hold without special finishing. PML01 (approximately 15cm x 15cm, depending on current Ethicon naming) is the standard size used for open inguinal hernia repair in the Lichtenstein technique, parastomal hernia reinforcement, and ventral hernia repair where the mesh dimensions suit the defect size. The approximate thickness is 0.020 inches (approximately 0.5mm). For surgical device distributors supplying hernia surgery programmes, Prolene Mesh is a consistently ordered product alongside the PROLENE hernia system pre-configured products. Sara Wellness exports Ethicon surgical devices and mesh products to medical distributors internationally.
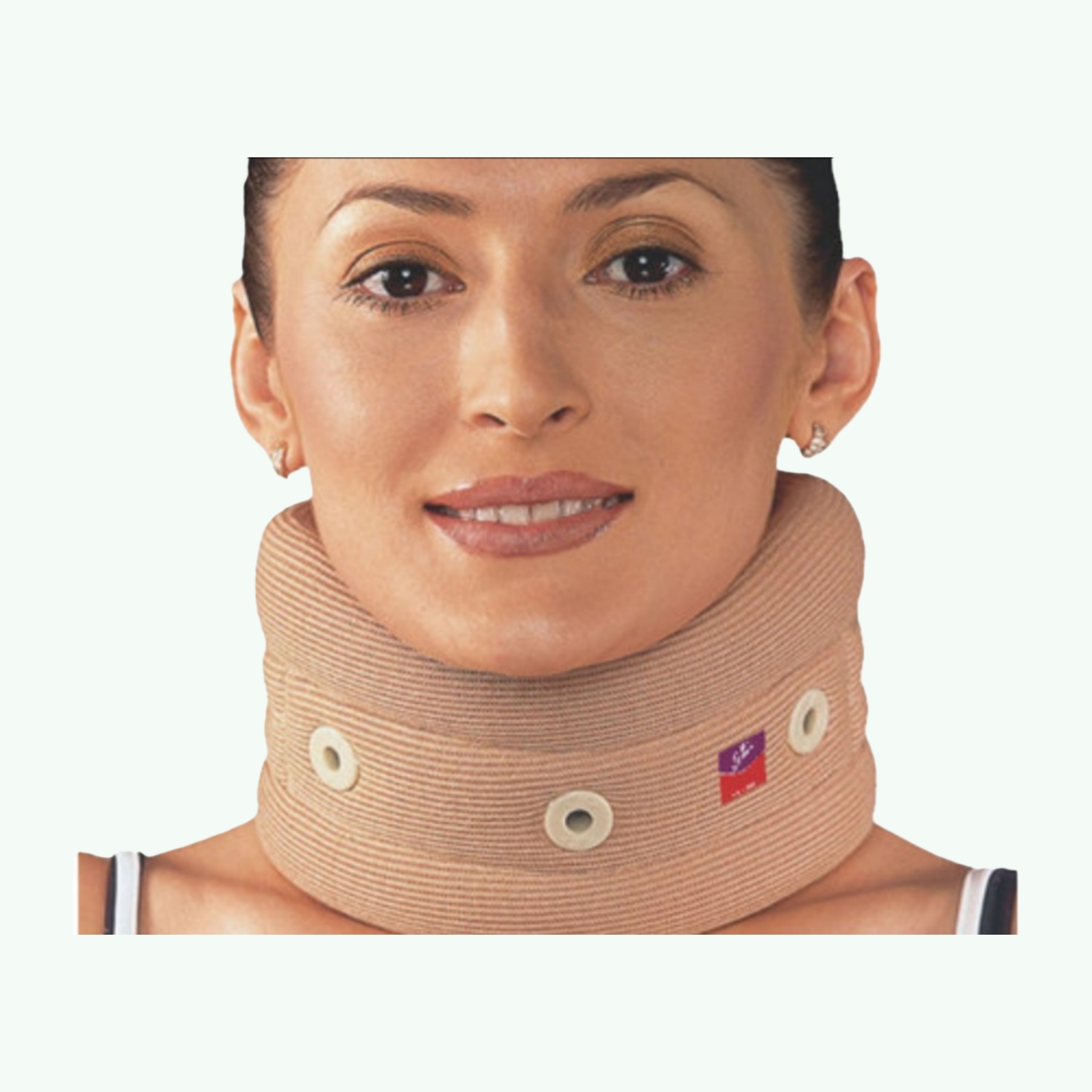
Flamingo Cervical Collar (Foam, Mild Support)
Few orthopaedic supports are prescribed as routinely as the soft foam cervical collar, and few are misunderstood as consistently. Patients receive them after road traffic accidents for whiplash assessment, after cervical spine surgeries as a post-operative aid, and for the management of cervical spondylosis pain — and in each case, the collar is serving a slightly different purpose. Understanding what a foam collar actually does, and what it does not, is relevant both for the clinical teams prescribing it and for the distributors sourcing it for their healthcare client base. Flamingo Cervical Collar is a soft foam collar made from closed-cell polyurethane foam covered with a cotton knitted stockinette fabric. This design provides partial immobilisation — it limits the most extreme ranges of cervical motion and provides a gentle physical reminder to the patient not to attempt rapid or extreme neck movements, but it does not achieve the rigid immobilisation that a hard collar or a Philadelphia collar provides. The clinical indication is therefore mild support, not complete cervical immobilisation. Where the Flamingo foam collar is genuinely valuable is in the kinesthetic reminder function: after whiplash injury or cervical strain, patients often inadvertently perform movements that aggravate their injury during the early recovery phase because they have not yet learned to protect the cervical spine from loading. The collar's presence provides constant proprioceptive feedback that keeps the patient aware of their neck position and encourages protective movement patterns during the critical first few weeks of recovery. The chin support contour accommodates the mandible without pressure, and the Velcro rear closure allows the collar to be adjusted for height and circumference fit. Air vents in the foam reduce heat buildup during extended daily wear. Available in S through XXL to accommodate the full range of adult neck dimensions. Flamingo collars are manufactured by Ascent Meditech, with a documented export presence across more than 60 countries. Sara Wellness exports Flamingo rehabilitation products to distributors internationally.
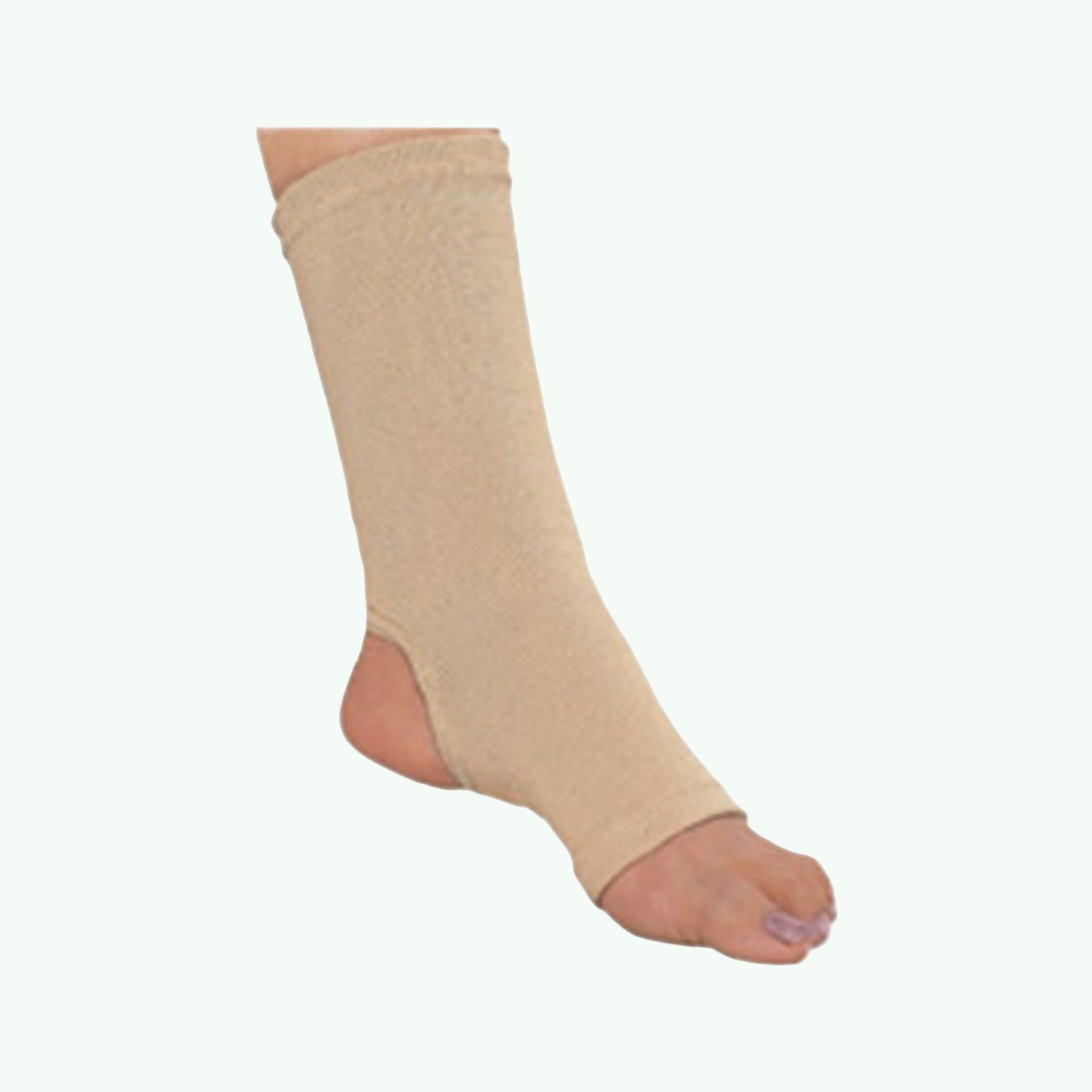
Flamingo Anklet Support Brace
Ankle sprains are among the most common musculoskeletal injuries in both athletic and everyday populations. The lateral ankle — specifically the anterior talofibular ligament — is vulnerable every time a foot rolls inward on an uneven surface, and the sprain that results ranges from a minor stretch requiring a few days of rest to a significant ligamentous disruption requiring weeks of structured rehabilitation. Somewhere in that range sits the majority of clinical ankle sprain presentations: painful, limiting, but not requiring surgical management. What they do require is support during the healing process. Flamingo Anklet Support Brace is an orthopaedic ankle support from Ascent Meditech Limited, designed to provide compression and mild mechanical support to the ankle joint during the recovery phase from sprains, strains, and mild instability. The brace is constructed from breathable elastic or neoprene-blend fabric, pulling on like a sock to position over the ankle joint, with compression that is distributed around the entire ankle circumference rather than concentrated at any single point. Some configurations include a silicone pressure pad positioned over the lateral malleolus — the bony prominence on the outer ankle — which provides targeted cushioning at the site most commonly involved in lateral ankle sprains, reducing impact irritation at the injury site during weight-bearing. The compression provided by the Flamingo ankle support serves to reduce post-sprain oedema, provide proprioceptive sensory input that helps the patient avoid extremes of ankle motion during healing, and reduce the experience of instability that makes the injured ankle feel unsafe for weight-bearing activity. The support is designed for use during the sub-acute and rehabilitation phases of ankle sprain recovery — once acute swelling has stabilised and partial weight-bearing has begun — rather than for acute injury immobilisation. For medical distributors, physiotherapy supply companies, and sports medicine supply chains, ankle support braces are a consistently purchased item at high volumes across consumer, clinical, and sports medicine channels. Sara Wellness exports Flamingo rehabilitation products internationally.
Flamingo Silicone Gel Heel Pads (Pair)
The heel absorbs approximately 110% of body weight with each step during normal walking gait — more during running. Over the course of a day of normal activity, this amounts to hundreds of thousands of Newton-seconds of compressive force directed through the calcaneus and into the surrounding plantar fat pad. When that fat pad atrophies (as it does progressively with age) or when a heel spur develops at the calcaneal insertion of the plantar fascia, or when plantar fasciitis inflames the fascia itself, the walking surface that used to feel neutral becomes a source of pointed pain with every step. Flamingo Silicone Gel Heel Pads are orthopaedic inserts designed to place a layer of viscoelastic shock-absorbing material between the calcaneus and the shoe's inner sole. The silicone gel material deforms under compression to distribute the impact of heel strike over a larger area and absorb a portion of the peak force before it reaches the heel's bony and soft tissue structures. The green-dotted centre section of many Flamingo heel pad designs concentrates the softest gel in the area of the heel that experiences maximum pressure, redirecting force away from the painful central heel area. The pad is designed to sit in the heel of a standard shoe or trainer without requiring any adhesive or modification to the footwear. The material is durable silicone that retains its gel properties through repeated compression cycles and can be switched between shoes — useful for patients who need to use the pad in their work shoe during the day and a different shoe for exercise. Clinical applications include plantar fasciitis heel pain, calcaneal heel spur pain, fat pad atrophy in elderly or diabetic patients, calcaneal bruising from impact injury, and preventive heel cushioning for patients who stand on hard floors for extended periods. For medical distributors, pharmacy chains, and physiotherapy supply companies, silicone heel pads are a high-volume, easy-to-stock item with a broad consumer base across age groups. Sara Wellness exports Flamingo foot care products internationally.
Flamingo Silicone Foot Insoles (Full-Length Gel Insole Pair)
The plantar surface of the foot is a complex load distribution system. The three-point contact of heel, first metatarsal head, and fifth metatarsal head forms the base of the foot's arch structure, and the entire soft tissue architecture of the plantar fascia, intrinsic muscles, and fat pads works to absorb and distribute the forces of walking across this structure. When any element of this system is disrupted — whether by flat foot collapse, metatarsalgia from forefoot overloading, diabetic fat pad atrophy, or post-surgical changes in gait — the result is pain and fatigue that affects the entire kinematic chain from the foot upward. Flamingo Silicone Foot Insoles are full-length orthopaedic insoles manufactured from medical-grade silicone gel, designed to provide whole-foot cushioning, arch support, and shock absorption for patients with a range of foot pain conditions. Unlike heel-only pads that address only the calcaneal pressure zone, full-length insoles redistribute plantar pressure across the entire foot surface, addressing metatarsal pain as well as heel pain. The full-length gel design cushions the ball of the foot — the metatarsal heads — where metatarsalgia pain originates, and provides a continuous shock-absorbing surface beneath the plantar fascia that reduces the repetitive strain forces that drive plantar fasciitis progression. The arch support element of the insole provides a medium of support beneath the medial longitudinal arch, counteracting the excessive arch flattening that occurs in patients with flexible flat feet and provides the arch drop that contributes to plantar fascia overloading. The insoles can be trimmed to fit specific shoe sizes using scissors — the trimming line is marked on the insole surface — making a range of sizes wearable from a single pair. For medical distributors, physiotherapy equipment suppliers, and pharmacy chains, full-length insoles are a higher-value product than heel pads alone, with a broad indication base across podiatric, diabetic foot, geriatric, and sports medicine patient populations. Sara Wellness exports Flamingo foot care products internationally.
Ethicon Vicryl Suture USP 0, 1/2 Circle Round Body (NW2346)
The volume of Coated VICRYL suture used in operating theatres around the world on any given day would be difficult to estimate with precision, but it would be very large. This is not an accident. It is the result of fifty years of clinical deployment across general surgery, obstetrics, gynaecology, urology, and orthopaedics, during which time the polyglactin 910 braided absorbable suture has proven itself reliable enough to become the default choice in surgical settings where an absorbable suture is appropriate. Coated VICRYL is a copolymer of 90% glycolide and 10% L-lactide, braided into a multifilament structure and coated with a mixture of polyglactin 370 and calcium stearate. The coating reduces surface friction, which makes tissue passage smoother and knot sliding more controlled than with uncoated braided absorbable materials. The braided structure provides the pliability and knot security that has made it preferred over monofilament alternatives in applications where multiple throws need to slide easily to the tissue surface. NW2346 is USP size 0 (EP 3.5), violet braided, 90cm strand, with a 40mm 1/2 circle round body needle. Size 0 on a round body needle covers a wide range of general surgery and obstetric procedures: uterine closure after caesarean section, bladder repair, bowel anastomosis, deep fascial layers, peritoneal closure, and subcutaneous tissue approximation in procedures where a single suture covers multiple steps. The 90cm strand is the standard length for continuous closure techniques — long enough to complete most single-layer closure runs without running short, without generating the excess material management issues that longer strands create in deep or narrow operative fields. For surgical supply distributors, Coated VICRYL in the size 0 round body configuration is among the highest-volume individual suture lines in any product catalogue. It is reordered at scale by virtually every hospital pharmacy and surgical supply chain that stocks Ethicon products. Sara Wellness exports Ethicon sutures internationally.
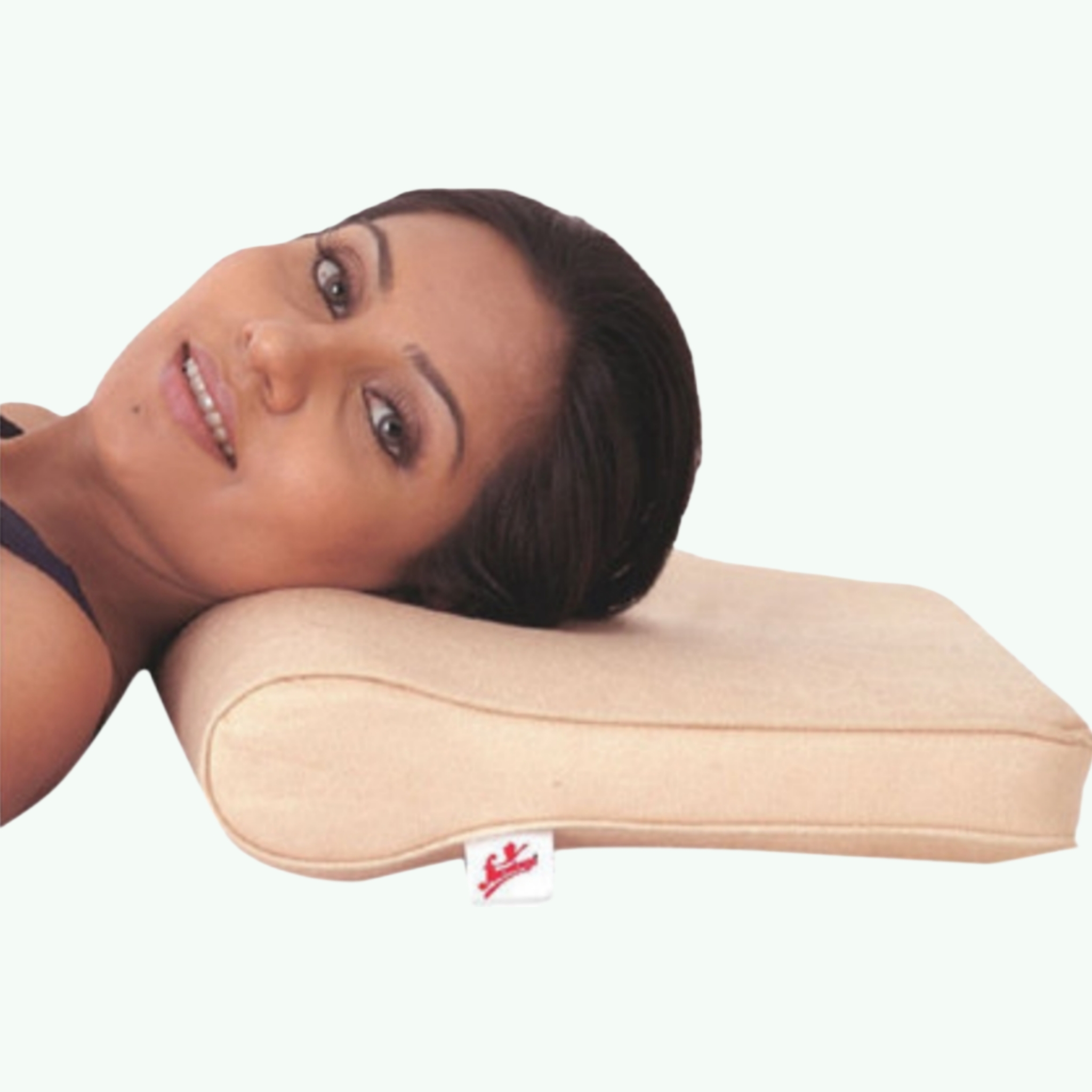
Flamingo Cervical Pillow (Orthopaedic Contoured Sleep Support)
Approximately one-third of a human life is spent sleeping. During those hours, the cervical spine is either supported in a position that allows the intervertebral structures to decompress and recover, or it is not. Most standard pillows make no commitment either way — they adapt to whatever position the sleeper adopts, which is often whatever position the shoulder width, mattress height, and habitual sleep posture dictate rather than whatever position the cervical spine would choose for itself. The Flamingo Cervical Pillow is an orthopaedic contoured foam pillow designed to maintain the cervical spine in neutral alignment during sleep. Neutral cervical alignment during sleep means the head is supported at a height and curvature that keeps the cervical lordosis in its natural position — neither flexed (head pushed forward by a pillow that is too high) nor extended (head falling backward against a pillow that is too low). In neutral, the musculature of the neck can relax without being stretched at end range, and the intervertebral discs and facet joints experience more even loading than in the deviated positions that standard pillows often produce. The contoured design typically features a lower central section and higher peripheral sections, with the geometry designed to support the head in the central depression while the curved edges support the neck during lateral rotation. Memory foam or high-density polyurethane foam variants of the Flamingo cervical pillow are available, with the foam density chosen to balance support firmness with pressure distribution. Flamingo cervical pillows are recommended by physiotherapists and orthopaedic surgeons for patients with cervical spondylosis, chronic neck pain, cervicogenic headache, and for post-operative cervical surgery patients who need to maintain specific sleep positions during recovery. For medical distributors, physiotherapy supply companies, and hospital supply chains supplying rehabilitation equipment, cervical pillows are a consistently recommended item with repeat purchase cycles tied to pillow wear and patient attrition. Sara Wellness exports Flamingo products internationally.
Ethicon Vicryl Plus Antibacterial Suture USP 1, 1/2 Circle Round Body (VP2350)
Post-operative wound infection is the variable that surgeons, infection control teams, and hospital administrators have been working to reduce for as long as there have been metrics to track it. Antibiotic prophylaxis covers the systemic side. Skin preparation covers the surface. What has traditionally received less attention is the suture surface itself — a several-centimetre length of braided synthetic material sitting in the wound, exposed to whatever bacteria are present in the operative environment, providing an interstice-rich surface that bacteria can use to establish biofilm before the host inflammatory response can clear them. Ethicon Vicryl Plus is Coated VICRYL (polyglactin 910) suture — the same braided absorbable suture that has been the standard for general soft tissue closure for decades — with the addition of triclosan coating. Triclosan is a broad-spectrum bacteriostatic agent that inhibits bacterial fatty acid synthesis, preventing bacteria from establishing colonisation on the suture surface during the first critical post-operative weeks. VP2350 is USP size 1 (EP 4), violet braided, with a 36-40mm 1/2 circle round body needle and a 90-120cm strand length — a large, heavy suture format designed for major fascial closures, uterine wall closure in caesarean section, bowel anastomosis, and deep abdominal tissue approximation where both tissue support and infection risk management are clinical priorities. The suture maintains the Vicryl absorption profile: 75% tensile strength at 14 days, 50% at 21 days, 25% at 28 days, with complete mass absorption between 56 and 70 days. The braided structure provides the excellent handling and knot security that has made Vicryl the high-volume standard for obstetric, general, and gynaecological surgery. Clinical evidence for triclosan-coated sutures shows approximately 30% reduction in SSI risk across a large meta-analysis, with NICE (UK) recommending triclosan-coated sutures as part of the SSI prevention bundle. Sara Wellness exports Ethicon sutures to surgical distributors internationally.
Ethicon Prolene Suture USP 0, 1/2 Circle Round Body (NW846)
There are materials in surgery that earn their position not through novelty but through decades of consistent, documented performance. Polypropylene suture is one of them. In vascular surgery, cardiac surgery, hernia repair, and any application where the demand is for a suture that remains mechanically stable in tissue for the life of the patient, polypropylene has been the answer for over fifty years. It simply does not degrade. It does not weaken. It sits in tissue with minimal reaction and holds what it was placed to hold. Ethicon Prolene is polypropylene monofilament suture — dyed blue for visibility, with the smooth, slippery tissue passage that is characteristic of polypropylene's surface properties. The monofilament structure eliminates the interstices of braided sutures where bacteria can shelter, and the surface has low coefficient of friction which means tissue trauma during the suture pass is minimal. NW846 is the product code for USP size 0 (EP 3.5), blue monofilament, 90cm strand, fitted with a 30mm 1/2 circle round body taper-point needle. The round body needle — a tapered point pushing through tissue rather than cutting it — is the correct needle choice for soft tissue approximation and vascular anastomosis, where the structural integrity of the tissue around the suture path matters. Size 0 is in the medium-heavy end of the Prolene range, suitable for general abdominal fascia closure, muscle layer approximation, hernia mesh fixation, and the majority of vascular anastomotic work outside the finest microsurgery. The 90cm working length is a considered design decision: long enough for continuous suture techniques in most procedures without creating a management problem, short enough that the suture does not tangle or stretch excessively during deep pelvic or thoracic work. For surgical suture distributors, Prolene in the size 0 round body format is one of the most consistently ordered polypropylene suture configurations in any product catalogue. Sara Wellness exports Ethicon sutures to medical distributors internationally.

Flamingo Abdominal Support Belt (Post-Surgery & Postpartum)
The first time a post-operative patient tries to sit up in bed after an abdominal procedure, they discover something no one quite prepares them for: the muscles that make sitting up feel effortless are the same ones that just went through surgery. Every cough, every laugh, every sneeze that was previously automatic becomes a moment of careful physical negotiation. This is where the abdominal support belt earns its place — not as a fashion item, not as a passive compression device, but as an active participant in the recovery process. Flamingo Abdominal Support Belt is manufactured by Ascent Meditech Limited, one of India's established orthopaedic soft goods manufacturers with a product range covering over 400 SKUs distributed across 60+ countries. The abdominal belt is their post-surgical and postpartum support solution, designed for the recovery phase following procedures like caesarean section, bariatric surgery, exploratory laparotomy, and hysterectomy, as well as postpartum recovery after normal delivery. The belt provides mild compression to the abdominal region through a broad elastic band that wraps around the midsection. Compression serves multiple clinical purposes in the recovery context: it reduces post-operative oedema by providing external pressure that limits fluid accumulation in the wound and surrounding tissue; it provides proprioceptive support to the abdominal muscles during movement, allowing earlier ambulation with less pain; and it acts as a physical reminder to the patient to engage their core during movement rather than relying purely on the injured abdominal wall. The construction uses breathable elastic that allows ventilation during wear, with semi-rigid internal stays that prevent the belt from rolling up at the edges — a common failure mode in cheaper abdominal supports that negates the compression benefit at the moment it is most needed. Velcro or hook-and-loop fastening allows easy adjustment and removal without requiring the patient to perform fine motor tasks that may be difficult post-operatively. Available in 14cm width (for lighter compression and narrower application) and 20cm width (for broader coverage and greater support of the lumbar-abdominal region), in sizes S through XXXL. Sara Wellness exports Flamingo rehabilitation products to medical distributors internationally.
G Surgiwear Turp Urological Drape
TURP — transurethral resection of the prostate — is one of the most commonly performed urological surgeries worldwide. It is also one of the highest-fluid procedures in urology. Irrigation volumes during resection can exceed several liters per case, and the draping system has to handle all of it without failure. The G Surgiwear TURP Urological Drape is specifically designed for this environment. The drape provides complete lithotomy-position coverage appropriate for endoscopic urological procedures, with a fluid collection pouch integrated into the design to capture irrigation runoff at the operative site. No pooling on the table. No soaked towels. A controlled, clean field from start to finish. Constructed from spunlace non-woven fabric with water-repellent treatment, meeting Level 4 barrier protection per ANSI/AAMI standards, the drape maintains its integrity through extended procedures. The collection pouch is positioned and sized for practical use with standard urological resectoscopes and irrigation setups. For urology departments running high TURP volumes, the economics of this drape are simple. For distributors supplying urology centers across international markets, the demand is consistent and the reorder cycle is short. Sara Wellness exports urological drapes and consumables from India. G Surgiwear has been manufacturing urological products and supplying hospitals internationally for over 30 years. We bring both the manufacturer relationship and the export experience to every order. Packs of 5, flexible MOQs, direct pricing.

Surgiwear Shunt Drape (Model D203)
Hydrocephalus shunt placement is neurosurgery. The field requirements are correspondingly exacting. This is not a procedure where a drape that lifts at the corner, passes fluid, or sheds fibers into the field is acceptable in any definition of the word. The Surgiwear Shunt Drape Model D203 is manufactured by the same company whose hydrocephalus shunts treat approximately one quarter of the world's shunt-dependent patients. G Surgiwear does not make a drape for shunt procedures as an afterthought. They make it because they understand the procedure, they supply the hospitals that perform it at scale, and they know exactly what a scrub nurse needs when that drape goes on. The D203 is constructed from spunlace non-woven fabric in the 65-77 GSM range, water-repellent, Level 4 protection per ANSI/AAMI standards. No fluid through the operating area. Minimum fiber transfer into the wound. No reactive chemicals in the fabric. For neurosurgical supply distributors and hospital procurement teams, the D203 is a specialist drape from a manufacturer with a genuine track record in shunt surgery. It comes in packs of 5. Sara Wellness exports this product as part of the wider G Surgiwear neurosurgical and draping range. We have the product knowledge, export documentation experience, and direct manufacturer relationship to support your procurement without the delays and uncertainty that come with going through multiple intermediaries.

G Surgiwear Surgiclude Orthoptic Eye Patches
Every parent who has tried sticking an eye patch on an unwilling six-year-old understands the problem with most orthoptic patches on the market: they either fall off by lunch or leave a red ring on the child's face by dinner. Usually both. The Surgiclude Orthoptic Eye Patch from G Surgiwear addresses both failure modes directly. This is a hypoallergenic adhesive eye patch with an absorbent non-stick pad, designed for managing strabismus (lazy eye) through occlusion therapy. The backing material is breathable — the kind of breathability that lets a child wear this to school without turning the experience into a battle. The gentle adhesive grips well enough to stay put through a normal day of activity while releasing cleanly at removal without pulling skin. The individual patch includes a non-stick absorbent inner pad that sits against the closed eyelid. No adhesive touches the eye area directly. The ergonomic oval shape is sized so the narrow end points toward the nose, following natural facial contours rather than fighting them. Two sizes: Small (63mm x 44mm) for pediatric use, Large (78mm x 55mm) for adult coverage or children needing fuller occlusion. Both come in boxes of 10 patches. Both are also used post-surgically when protective eye coverage is needed for healing without the clinical weight of a full eye dressing. G Surgiwear has supplied ophthalmic products to hospitals and clinics for over three decades, certified under EN ISO 13485. Sara Wellness exports this and the full G Surgiwear ophthalmic range from India. We have been at this for 15 years, we know how to pack for international transit, and we respond to emails within 24 hours. That last part is rarer than it should be.

G Surgiwear Shoulder Arthroscopy Drape (Model D509)
Shoulder arthroscopy generates fluid. A lot of it. Irrigation fluid, joint fluid, blood — it goes everywhere during the procedure, and the drape system is the last line of defense between all of that and the sterile field, the patient, and the OR floor. The G Surgiwear Shoulder Arthroscopy Drape Model D509 was developed with direct input from orthopaedic surgeons who operate in exactly this environment. The drape is constructed from 65-77 GSM spunlace water-repellent fabric. Two sheets: 160x190cm and 160x210cm. The structured construction delivers Level 4 protection as per ANSI/AAMI standards. No fluid passes through the operating area of this drape. The fabric transfers minimal fibers into the wound. There are no chemicals present in the drape material that could react with wound tissue. For surgeons and scrub nurses who manage shoulder cases, this translates to a dry, stable, cleanly draped field from incision to closure. For infection control teams, it means one fewer variable in post-operative complication prevention. The D509 is available in packs of 5. For orthopedic surgery centers running high shoulder arthroscopy volumes, the economics of pack-of-5 sterile drapes are straightforward to calculate. G Surgiwear manufactures and exports to over 50 countries. Sara Wellness handles the India-to-world logistics for buyers who need consistent supply, accurate documentation, and a contact person who actually picks up the phone. We have been doing this for 15 years. We know what paperwork your customs authority needs.

G Surgiwear Solid Synthetic Testis Testicular Implants
Orchidectomy — surgical removal of a testicle — is performed for testicular cancer, severe trauma, torsion, and other conditions. What follows the surgery is a period of physical recovery that is well understood. What is less often discussed is the psychological dimension of the absence, and how a properly placed testicular prosthesis can address that dimension quietly and effectively. The G Surgiwear Solid Synthetic Testicular Implant is a solid silicone prosthesis designed to restore natural scrotal appearance and feel following unilateral orchidectomy. The solid construction means no fluid-filled chambers, no valve mechanics, and no risk of deflation or rupture over time. The device maintains its form and feel indefinitely under normal physiological conditions. G Surgiwear has been manufacturing andrology implants alongside their broader range of neurosurgical, orthopaedic, and urological products for over 30 years. The testicular implant, while a lower-volume product than their shunts or orthopaedic devices, is manufactured to the same quality standard: EN ISO 13485:2012 certified, exported internationally, and available through specialist medical distributors worldwide. Available in small, medium, and large sizes to accommodate patient anatomy. Each implant is supplied sterile, ready for surgical placement. Sara Wellness exports this and related G Surgiwear urology products from India. We work with medical distributors, hospital chains, and specialty surgical supply companies. Regulatory documentation for device importation can be arranged. Tell us where you are importing to and what you need, and we will handle the rest.

G Surgiwear Penile Implant
Andrology implant surgery is a field where the margin for error is essentially zero. The patient population, the psychological stakes, and the permanence of the procedure demand a device that performs without compromise for years after implantation. G Surgiwear's penile implant is a semi-rigid malleable prosthesis from one of India's most established implant manufacturers. The company's implant engineering heritage runs deep — they are the same manufacturer responsible for a significant portion of the world's hydrocephalus shunt supply. Their experience with implantable devices is not a marketing claim. It is measurable in procedure volumes and three decades of post-market history. The semi-rigid design allows the implant to be manually positioned for intercourse and repositioned to a non-erect configuration for daily wear. It does not require a pump, reservoir, or mechanical inflation cycle, which means fewer mechanical components and fewer mechanical failure modes. For patients in markets where healthcare access for complex mechanical revisions is limited, this matters enormously. Manufactured under EN ISO 13485:2012 certification and exported to multiple countries, this device is supported by the manufacturing rigor expected of a serious implant product. Sara Wellness has 15 years of experience exporting G Surgiwear implants and surgical products to international markets. We understand the documentation requirements for Class III device importation, we know the regulatory landscape in multiple regions, and we deal with importers directly. If you are sourcing andrology implants for a distributor network or hospital chain, this conversation is worth having.

Surgiwear PCNL Drape Eco (Model E013)
Percutaneous nephrolithotomy is not a gentle procedure. The patient is prone, the kidney is accessed through the back, and the irrigation volumes are substantial. The drape system handling that case needs to manage large amounts of fluid without failure — quietly, reliably, every single time. The Surgiwear PCNL Drape Eco Model E013 is built specifically for this scenario. PCNL drapes are a specialized category within urological draping. The E013 provides full sterile coverage for prone PCNL positioning, with a construction that handles the fluid-heavy environment of nephroscopic access. Level 4 protection per ANSI/AAMI means the drape resists fluid penetration in the operating area, which is the critical requirement for procedures that run long and wet. The Eco designation indicates this is a cost-optimized variant within the G Surgiwear PCNL drape range, providing necessary barrier performance at a procurement price that works for hospitals running high procedure volumes. The performance where it matters does not change. The material is still spunlace non-woven with water-repellent treatment, consistent across the G Surgiwear drape line. For urology supply distributors and hospital procurement teams sourcing PCNL-specific draping solutions, the E013 is a direct import option from India with a manufacturer who has been supplying these products internationally for over three decades. Sara Wellness handles the export side of this. Fifteen years of experience, documentation sorted, shipment tracking provided, and direct contact throughout the process.

Surgiwear Eye Drape Plain (Model D700AP)
Cataract surgery volumes are climbing worldwide. More phacoemulsification procedures. More day-care ophthalmic theatres. More need for a reliable, consistently performing ophthalmic drape that does not complicate a procedure that already demands total precision. The Surgiwear Eye Drape Plain Model D700AP is the disposable answer to that demand. Designed specifically for ophthalmic surgery, the D700AP creates a sterile field around the eye and face. The drape features an appropriately positioned fenestration that aligns with the surgical site, giving the surgeon clean, unobstructed access to the operative eye while maintaining complete coverage of surrounding non-sterile surfaces. Manufactured in 65-77 GSM spunlace non-woven fabric, the material is fluid-repellent and meets ANSI/AAMI Level 4 barrier standards. This means it does what ophthalmic drapes must do: stay dry, stay in place, and not shed fibers into the surgical field where any contamination is a catastrophic problem. G Surgiwear supplies ophthalmic drapes to hospitals across 50 countries. They have been manufacturing ophthalmic and surgical disposables for over 30 years from their ISO-certified facility in Shahjahanpur. Sara Wellness exports their products and others in this category from India. We work with ophthalmology supply distributors, surgical chains, and government hospital procurement departments. Tell us your volumes and destination, and we will come back with a practical number.

G-Dress Swim Proof Sterilized Adhesive Bandage
The patient showers. Gets sweaty. Goes back to work two days after the procedure. And they absolutely will not stop moving no matter how firmly you explain the concept of wound rest. The G-Dress Swim Proof Adhesive Bandage was made for that patient. G-Dress is a ready-to-use, sterilized adhesive wound dressing built on an SMS (Spunbond-Meltblown-Spunbond) non-woven laminate. The middle meltblown layer is the key piece. It resists fluid passage, which is what makes this dressing water-resistant and what keeps shower water and pool water on the outside where they belong. The wound stays protected. The dressing stays in place. Available in Size 5 (standard pack of 25) and a transparent-with-pad variant (pack of 10), G-Dress covers most dressing scenarios from routine wound coverage to active-patient post-operative care. The transparent variant allows visual wound assessment without disturbing the dressing, which the clinical team appreciates. The patient appreciates not having to come back for a dressing change every 24 hours. For wholesale buyers and distributors sourcing wound care consumables from India, G-Dress represents the kind of product that moves consistently. Every surgical ward, every outpatient clinic, every patient who refuses to stay dry. The demand does not stop. Sara Wellness exports wound care products and surgical dressings from India. Fifteen years, many markets, and a track record of getting shipments to port on time and documentation correct on the first attempt. Get in touch and we will sort out your requirements.

G Surgiwear Eye Pad
Cotton inside. Filament yarn outside. Sealed on all four sides. The G Surgiwear Eye Pad sounds like a simple product, and that is precisely the point. In ophthalmic care, the fibers that contact a healing eye cannot be loose. They cannot shed. They absolutely cannot migrate into the surgical site. Older eye dressings made of plain gauze fail this requirement quietly and often — you just do not see the problem until complications appear. This eye pad was designed to make that failure mode impossible. The pad construction encases cotton inside a knitted filament yarn fabric sealed on all sides. No fiber protrudes. None. The inner cotton handles absorption. The outer shell handles everything else, including the parts that touch the patient. The adhesive patch securing the pad in place is cut from soft, stretchable fabric with a gentle adhesive that is easy to remove without distressing periorbital skin. The release liner peels cleanly, which matters when the patient is anxious, the assistant is gloved, and time is limited. This comes in standard and economical variants. Both are sterilized using ethylene oxide. Pack of 50 units per pack, individually packed in peel-open inner pack with a tear-open outer pack — designed for the sterile field. For importers sourcing ophthalmic consumables at volume, the G Surgiwear Eye Pad sits at the dependable middle of the market. It is not the cheapest option in the category. It is the one that works without complaint, procedure after procedure. Sara Wellness exports this product along with a broad ophthalmic consumables range from India. We have been doing this for 15 years, and we negotiate on MOQs without making it a 12-email process.