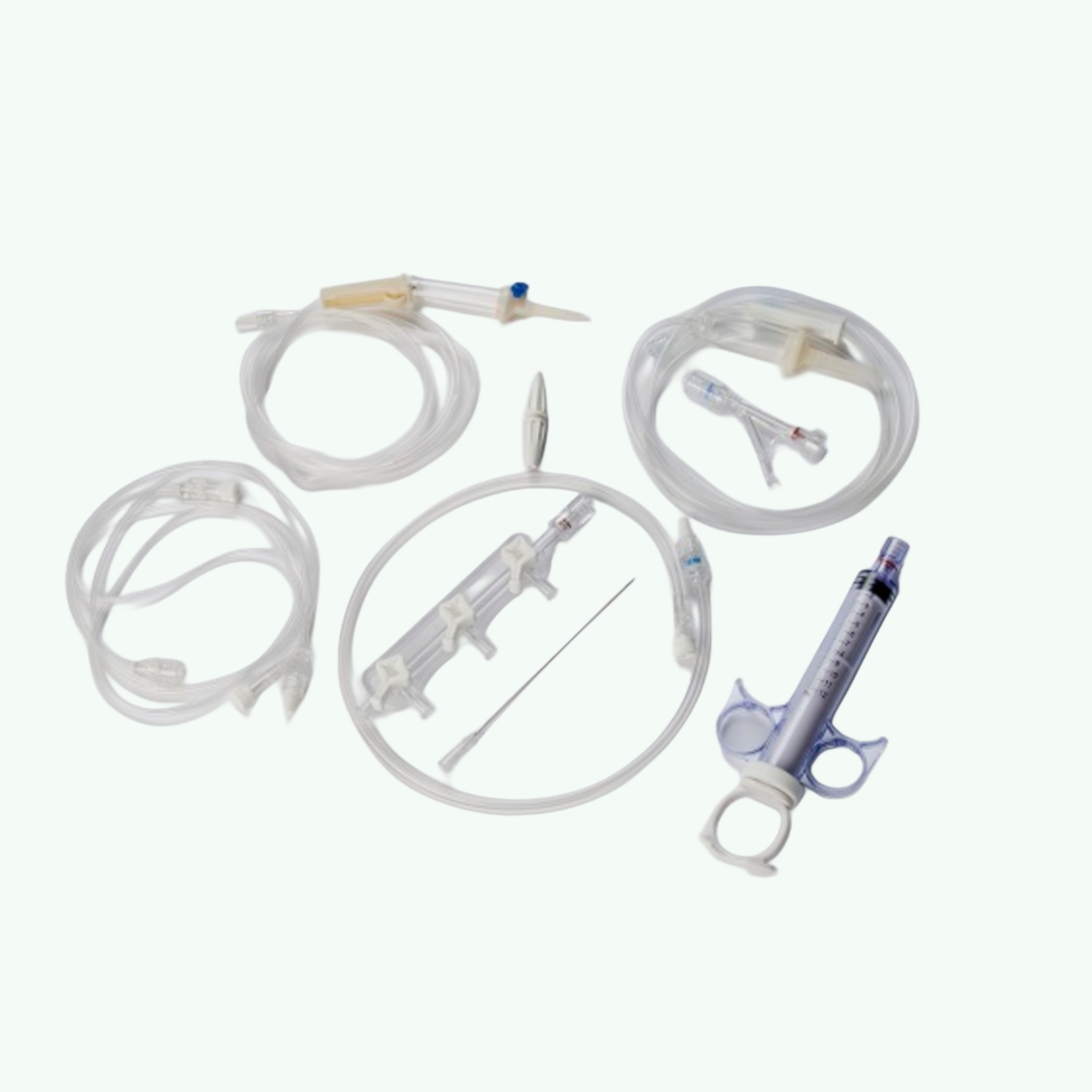
Clearkit PTCA Kit (Angiography Kit)
Pack Sizes Available
Product Description
Technical Specifications
- Procedure Type: Coronary angiography and PTCA (percutaneous transluminal coronary angioplasty)
- Access Options: Femoral artery or radial artery (transradial) configuration
- Key Contents: Introducer sheath, dilator, guidewires, manifold, connecting tubing, stopcocks, syringes, ancillaries
- Sterility: Sterile, single-use disposable components; ETO sterilised
- Customisation: Kit configurations can be adapted to match facility-specific procedure protocols
- Regulatory: CE marked; complies with applicable medical device standards
Frequently asked questions
A PTCA angiography kit typically contains a vascular access introducer sheath with dilator, guidewires (standard and/or J-tip), a manifold with pressure ports and stopcocks, connecting tubing, Y-connectors, three-way stopcocks, syringes of various sizes, procedural needles, and ancillary items such as patient drapes and surgical swabs. The exact configuration varies by kit design, access site (femoral or radial), and the facility's procedure protocol.
A diagnostic angiography kit is used for coronary angiography — visualising the coronary arteries with contrast injection — without intervention. A PTCA kit is designed for interventional procedures that include balloon dilation of a narrowed coronary artery. The interventional kit includes all the components of the diagnostic kit plus components for catheter exchange, balloon delivery, and stent deployment. Specific interventional components (balloons, stents) are typically added separately based on the specific lesion characteristics.
PTCA kits are available configured for femoral artery access (the traditional approach via the groin) or radial artery access (transradial, via the wrist). Radial access has become increasingly preferred due to lower access site complication rates and earlier patient ambulation. The introducer sheath diameter and guidewire specifications differ between approaches. Specify the preferred access site when ordering kit configurations.
Yes. All components in a PTCA angiography kit are sterile, single-use disposables. They are intended for use in a single procedure on a single patient and must be discarded as clinical waste after use. No component should be reprocessed or reused. The sterile packaging maintains sterility until the point of use, and kits should be inspected for packaging integrity before opening.
Yes. PTCA procedure kit manufacturers and distributors commonly offer kit customisation to match hospital or cathlab-specific protocols. Components that a facility does not use can be removed, and additional items specific to a facility's procedure preference can be added. Custom configurations reduce waste, ensure every required component is present, and simplify inventory management. Contact the supplier with the facility's procedure protocol for a configured kit proposal.

Introducer Sheath (Vascular Access Sheath)
Vascular access in catheterization procedures operates on a principle that seems simple until you watch a novice struggle with it for the first time. Get a catheter into a vessel without tearing the vessel wall, maintain hemostasis around the catheter, allow catheter exchanges without losing access, and do all of this through a puncture site the diameter of a pencil lead. The introducer sheath exists because attempting those maneuvers without one turns a five-minute procedure into a twenty-minute ordeal with multiple access attempts. Introducer sheaths are the foundational vascular access device that makes modern interventional cardiology, radiology, and electrophysiology possible. This is a thin-walled tubular device inserted percutaneously into arteries or veins to provide a stable conduit for repeated introduction, manipulation, and exchange of catheters, guidewires, and interventional devices during diagnostic and therapeutic procedures. The sheath consists of a radiopaque polyurethane or PTFE tube with an integrated hemostasis valve at the proximal end that maintains seal around inserted catheters while minimizing backbleeding and air aspiration. The side port with attached three-way stopcock allows pressure monitoring, blood sampling, or fluid/medication infusion without disturbing the main catheter pathway. French sizing matters because it determines which devices fit through which sheaths. The sheath size refers to inner diameter while catheter size refers to outer diameter, which is why a 6Fr catheter fits through a 6Fr sheath. The sheath's outer diameter is typically 2Fr larger than its nominal size due to wall thickness. Understanding this sizing convention prevents the frustration of attempting to insert an 8Fr device through a 7Fr sheath and wondering why physics suddenly stopped cooperating. The dilator tapered tip facilitates initial insertion over a guidewire through the vessel wall puncture. Once the sheath-dilator unit is seated in the vessel, the dilator and guidewire are removed, leaving the sheath in place as a working channel. The SnapLock feature on many modern sheaths secures the dilator during insertion, preventing separation that would compromise the insertion. Hemostasis valve technology has evolved considerably. Modern valves maintain seal integrity across a range of catheter sizes, allow smooth catheter manipulation without excessive friction, and preserve catheter torque transmission characteristics that are critical for steering in complex anatomy. Poor valve design either leaks profusely or grips so tightly that catheter manipulation becomes impossible. Sheath lengths are procedure-specific. Short sheaths (5-7cm) are used for radial artery access where the working distance to target anatomy is short. Standard sheaths (10-11cm) accommodate femoral access for most coronary and peripheral procedures. Long sheaths (20-30cm) are required for complex anatomies, transseptal access, or when the working catheter needs support deep in the vasculature. For distributors supplying catheterization laboratories, interventional radiology suites, and electrophysiology labs, introducer sheaths represent essential consumables with predictable usage tied directly to procedural volume. Sara Wellness has been exporting interventional cardiology and cathlab supplies from India for 15 years.

ClearCath Coronary Diagnostic Catheters
Cardiology cath labs run on precision. And when hospitals, distributors, and procurement teams across the globe go searching for coronary diagnostic catheters in bulk, what they're really asking is: can we trust this supplier to get it right, every single shipment, no surprises? That's the question Sara Wellness has been answering for over 15 years. ClearCath coronary diagnostic catheters are preformed, single-use catheters designed for diagnostic coronary angiography. The range covers the shapes that cath labs actually need day to day — Judkins Left, Judkins Right, Amplatz Left, Amplatz Right, Pigtail, and Multipurpose — available in 4 Fr, 5 Fr, and 6 Fr sizes at the standard 100 cm working length. Whether your market leans femoral or is increasingly going radial, the range has you covered. Think of it this way: a cath lab can't afford a catheter that loses torque mid-procedure, or one whose tip doesn't hold its preformed shape under pressure. ClearCath catheters are built to maintain shape memory and deliver predictable handling, which is exactly what interventional cardiologists expect. Each unit is sterile packed individually, ready for the OR tray. Now, here's the part that matters most to importers and wholesale distributors. You want a supplier who doesn't disappear after the proforma invoice is signed. With Sara Wellness, every order gets a dedicated point of contact, not a rotating inbox. We talk through your French size mix, your shape requirements, your country's import documentation, and your delivery timelines — before the order is placed, not after something goes wrong. We've supplied cathlab consumables to buyers across Africa, the Middle East, and Southeast Asia, and our pricing reflects a genuine understanding of what these markets can sustain. No inflated quotes followed by mysterious discounts. Just transparent, competitive pricing from the start. Minimum order quantities are flexible, especially for first orders where you're evaluating a new supplier. That's a deliberate policy, not a sales tactic. We'd rather you order a test quantity, verify the product quality in your own cath labs, and come back for volume — than push you into a container load on the first conversation. If you're a medical importer, a hospital procurement manager, or a regional distributor looking for a reliable Indian exporter of coronary diagnostic catheters, let's have a real conversation. The paperwork will be clean, the shipment will be on time, and the product will do exactly what it's supposed to do inside a cath lab.
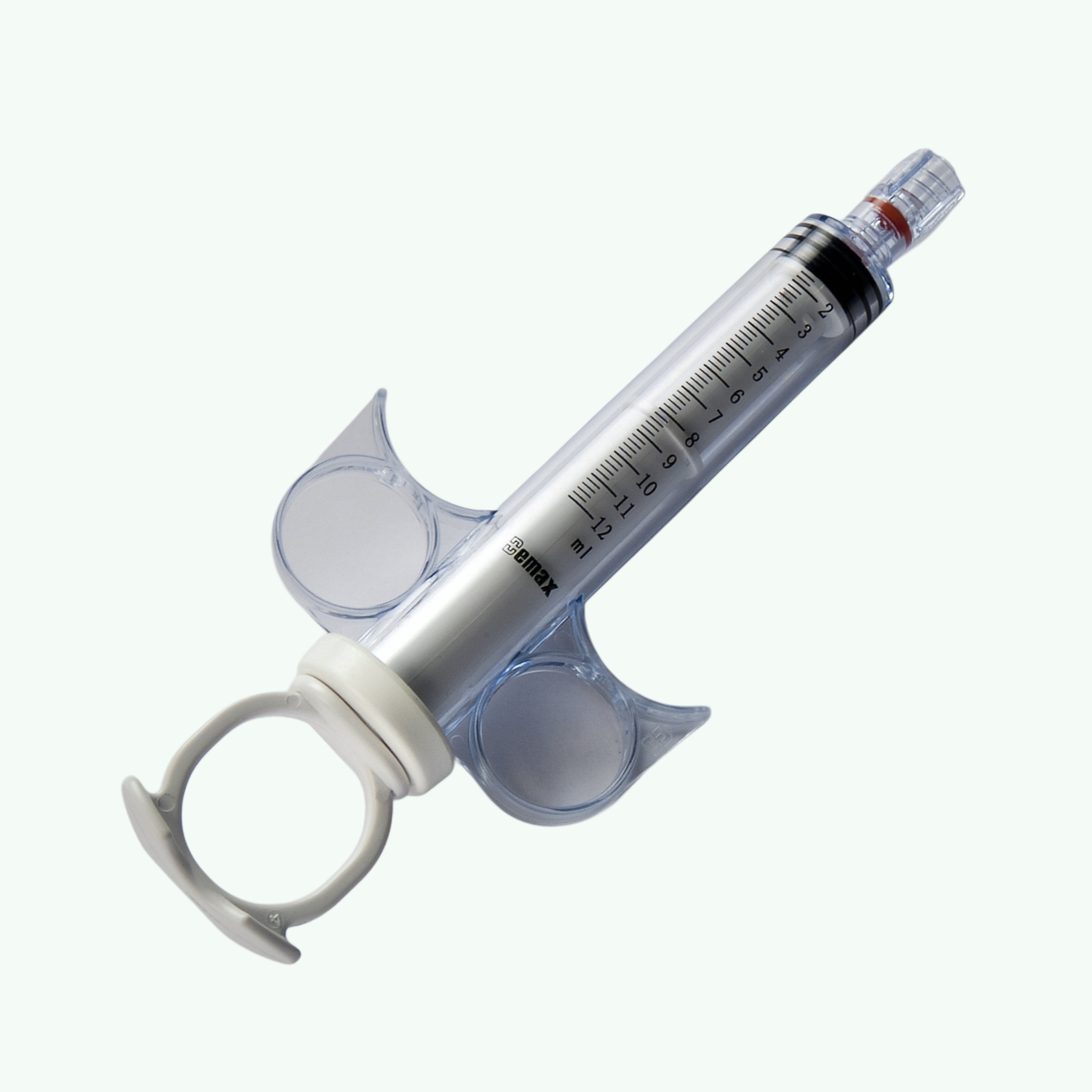
Coronary Control Syringe
In a cathlab, control is everything. Precision is not a luxury. It is the quiet expectation behind every successful coronary procedure. The Coronary Control Syringe is designed to offer clinicians steady, controlled contrast injection during angiography and interventional cardiology procedures. For importers and distributors, however, the real question goes beyond design. It is about sourcing reliability, regulatory confidence, and supply continuity. That is where Sara Wellness steps in. We export coronary control syringes to hospitals, cardiac centers, and medical distributors across global markets. Each unit is manufactured for smooth plunger movement and controlled delivery, helping healthcare professionals manage contrast media administration with confidence. Clear graduation markings allow easy visibility, while ergonomic finger grips support stable handling inside demanding cathlab environments. But product quality alone does not build a long term partnership. Consistent documentation does. Timely dispatch does. Clear communication certainly does. With over 15 years in medical supplies export, we understand how cardiac consumables move across borders. We assist buyers with organized packing, proper labeling, and coordinated shipping schedules. Whether you are supplying government hospitals in Africa, private cardiac centers in the Middle East, or distributors in Southeast Asia, our team works closely with you to maintain predictable supply. Flexible minimum order quantities help emerging distributors enter new markets without excessive inventory pressure. Larger buyers benefit from structured bulk pricing designed to protect margins in competitive healthcare sectors. Every order is handled with direct, one on one coordination because in medical trade, assumptions are expensive. Our coronary control syringes are supplied sterile and individually packed, suitable for hospital procurement and resale distribution. Transit packaging is arranged carefully to maintain product integrity during long distance shipping. If you are searching for a dependable coronary control syringe exporter from India, Sara Wellness offers not just supply, but accountability. In cardiac care, there is no room for uncertainty. Your supply chain should reflect the same standard.